Acute Aortic Syndromes
Gila Perk
Itzhak Kronzon
ACUTE AORTIC SYNDROMES
The term “acute aortic syndrome” (AAS) has been used to describe a mixed group of thoracic aortic disorders that characteristically present with a typical combination of signs and symptoms.1,2,3 and 4 The most distinctive presenting symptom is aortic pain, which is often described as sharp, tearing, or ripping. Although there are several pathophysiologic entities that can present as AAS (Table 23.1) occasionally one process can precede another or occasionally they may coexist. Importantly though, all can be life-threatening and require immediate medical attention (often including a surgical intervention). Given the similarity in the clinical presentation and the need for urgent assessment, diagnosis, and treatment, it is useful to classify these syndromes into one group of disorders, similar to “acute coronary syndromes” or an “acute abdomen.”
AAS include the following:
Aortic dissection
Intramural hematoma, and
Penetrating aortic ulcer
Typically, patients with AAS present with an acute onset of chest pain with a long-standing history of hypertension or a known elastic tissue disorder (e.g., Marfan syndrome). The pain is classically described as abrupt in onset, intense, sharp, tearing, or ripping. Diseases that involve the ascending aorta typically produce pain that radiates to the neck or the jaw. Disease in the descending thoracic aorta often causes pain that radiates to the back or abdomen.5
A related process that will be discussed in this chapter as well is traumatic aortic rupture. Although aortic rupture is obviously a medical emergency that can present with symptoms of aortic pain similar to other AAS, patients present with a completely different scenario of onset of this process. For this reason, traumatic rupture is often not considered part of the classical AAS.
TABLE 23.1 Acute Aortic Syndromes | ||||
|---|---|---|---|---|
|
AORTIC DISSECTION
Aortic dissection is characterized by an intimal tear and separation of the intima or media from the surrounding adventitia. Typically, the separation occurs within the media such that the outer layer of the media forms the outside wall of the false lumen and the inner part of the media, along with the intima, forms the intimomedial flap. The thickness of the media that forms the external layer of the false lumen determines the stability of the false lumen wall and is an important determinant of the risk of free wall rupture. The dissection can propagate through the aortic wall both distally and proximally to the initial tear. This propagation can be the cause for many of the complications of aortic dissection (e.g., myocardial ischemia, renal failure). In many patients a reentrance tear can be found or multiple communications between the true and false lumens can be identified.
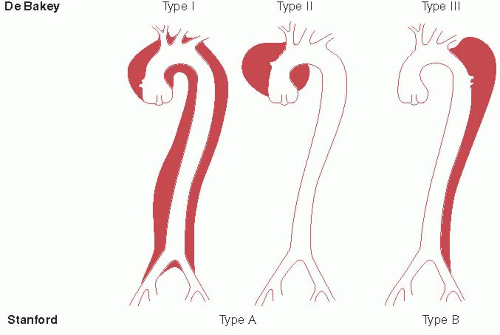
There are two widely accepted anatomic classifications of aortic dissection; the DeBakey and the Stanford classification (Figure 23.1). The DeBakey system divides aortic dissection to three subtypes: types I, II, and III. Type I DeBakey is a dissection that starts at the ascending aorta and continues at least up to or beyond the arch. Type II starts at the ascending aorta and is confined to the ascending aorta. Type III is a dissection that starts at the descending thoracic aorta and propagates either distally or less commonly proximally. The Stanford classification differentiates aortic dissection according to presence or absence of ascending aorta involvement, regardless of the initial site of origin; type A is a dissection involving the ascending aorta, and type B is a dissection that does not involve the ascending aorta. The Stanford classification is the one clinically used for treatment purposes; urgent surgical intervention versus medical stabilization. Because Stanford classification is the more commonly used system, we will use it throughout this chapter.
EPIDEMIOLOGY
The incidence of aortic dissection is estimated to be 2.9 per 100,000 patient-years. The mean age of presentation is 65 years with approximately 60% predominance of men.5,6 and 7 Women with acute aortic dissection present at an older age than men and tend to present later in the course of the disease than men.8 Type A aortic dissection is more common than type B and accounts for 60% of cases of acute aortic dissection. Up to 30% of patients with acute type A dissection show involvement of the aortic arch as well.
Similar to acute ischemic syndromes, a circadian as well as seasonal variation in the frequency of acute aortic dissection
has been found.9 Acute aortic dissection is significantly more prevalent between 6 a.m. and 12 noon, and in particular between 8 a.m. and 9 a.m. A seasonal variation in acute aortic dissection presentation has also been found, with more frequent occurrence in the winter as compared to summer, with a peak incidence in January.
has been found.9 Acute aortic dissection is significantly more prevalent between 6 a.m. and 12 noon, and in particular between 8 a.m. and 9 a.m. A seasonal variation in acute aortic dissection presentation has also been found, with more frequent occurrence in the winter as compared to summer, with a peak incidence in January.
Acute aortic dissection presents slightly different in North American and European adults.10 North American patients tend to be older, demonstrate more ECG changes (significantly more nonspecific ST changes and a trend towards more ST elevations and acute myocardial infarcts), and have more atypical symptoms and less classic chest X-ray findings compared with European patients. Despite the differences in clinical presentation, there is no difference in the early outcome between North American patients and European patients.
ETIOLOGY
The underlying pathology in many cases of aortic dissection is cystic medial necrosis causing degeneration of the aortic media. Of note, this is in fact a misnomer as pathology specimens do not demonstrate either cysts or necrotic material. However, it is a commonly used term that describes the generative process of the aortic media that involves degeneration of the elastic tissue. There are several predisposing factors that have been identified as risk factors for aortic dissection. The commonest risk factor that has been recognized is systemic hypertension. Up to 72% of patients who present with acute aortic dissection have a history of systemic hypertension.7 History of atherosclerosis is also common; it is found in up to 32% of patients presenting with acute aortic dissection. These risk factors are particularly noteworthy in patients above 40 years of age who present with acute aortic dissection.
Acute aortic dissection has also been found to be associated with collagen/elastic tissue diseases such as Marfan syndrome and Ehlers Danlos syndrome. Of all patients with acute aortic dissection, 4.9% have a history of Marfan syndrome.5,7,11 However, among patients younger than 40 years of age who present with acute aortic dissection, 50% have Marfan syndrome.11
Several other predisposing risk factors have been identified in patients presenting with acute aortic dissection5,7,11:
Prior aortic dilatation and aneurysm: Up to 16% of all patients (and 19% of patients younger than 40 years) presenting with acute aortic dissection have a prior history of aortic aneurysm.
Bicuspid aortic valve: Among young patients (<40 years old) who present with acute aortic dissection, 9% have a bicuspid aortic valve. Bicuspid aortic valve is frequently associated with progressive dilatation of the ascending aorta, irrespective of the valvular function. This is likely due to significant loss of elastic fibers in the aortic media.12 In patients with bicuspid valve-associate dissection, the ascending aorta is always involved (type A dissection).
Prior history of aortic dissection is found in 4% to 7% of patients with acute aortic dissection, and up to 11% of patients presenting with type B dissection.
Prior cardiac surgery is seen in up to 18% of patients presenting with acute aortic dissection. These include prior aortic surgeries (aneurysm or dissection repair) and other cardiac surgeries in which the aorta is surgically manipulated (such as coronary artery bypass grafting). Cardiac catheterization and percutaneous coronary interventions have been seen to precede in about 2% of cases of acute aortic dissection.
Somewhat conflicting evidence exists regarding the casual relationship between cocaine use and acute aortic dissection.13 In a multicenter, large international registry, cocaine use has been found seldom among patients presenting with acute aortic dissection (<1%). However, in an inner-city-based series, recent (minutes to hours before presentation) cocaine use has been reported in 37% of patients presenting with acute aortic dissection.14 The difference is most likely related to the different patient populations represented in these studies. Cocaine has been suggested to cause weakening of the aortic elastic media, premature atherosclerosis, and sudden onset of severe sheer forces (from the significant hypertension and tachycardia associated especially with crack cocaine use). The combination of these chronic and acute effects of cocaine use could be related to the risk of acute aortic dissection.
Inflammatory diseases involving the great vessels such as syphilitic aortitis, and vasculitis of the aorta (e.g., Takayasu vasculitis or giant cell arteritis) have been associated with aortic dissection in rare instances.
Pregnancy: There have been several reports of acute aortic dissection complicating pregnancy. Most have been associated with Marfan syndrome as well, and it remains unclear whether pregnancy represents an additive risk or whether it is merely a common enough condition that these associations may not represent a causative relationship.
CLINICAL MANIFESTATIONS
The hallmark of the clinical presentation of acute aortic dissection is “aortic pain.” This is frequently described as an abrupt onset of severe, sharp, tearing, or ripping chest pain. Radiation to the jaw or the neck is more common with type A dissection, whereas radiation to the back is seen more commonly with type B or abdominal aortic dissection. However, painless dissection has also been described, in up to 6.4% of patients presenting acute aortic dissection.15 Patients who present with painless dissection are older than those with painful dissection, have a higher prevalence of type A dissection, and present more commonly with syncope, congestive heart failure, and stroke. The mortality rate in this group is also higher as compared to those who present with acute aortic pain, in particular in the group with painless type B dissection (who have a significantly elevated risk of aortic rupture).
Patients with acute aortic dissection can also present with syncope; this has been described as the presenting manifestation in 13% of patients and was found to be associated with significantly increased in-hospital mortality.16 In multivariate analysis, syncope has been found to be associated with presence of cardiac tamponade, stroke, and proximal aorta involvement. The increased mortality risk associated with syncope persisted after controlling for patients’ demographic characteristics but not after adjusting for the comorbid conditions (stroke, tamponade, and proximal dissection).
Pulse deficit (absence of carotid, brachial, or femoral pulse owing to intimal flap involving major branch vessels or compressing hematoma) has been described in up to 15% of patients, more commonly in type A dissection versus type B (19% vs. 9%, respectively). Pulse deficit has been associated with increased in-hospital complications (such as renal failure, coma, hypotension, and limb ischemia) as well as an independent predictor of 5-day in-hospital mortality.17
Patients with acute dissection can present with hypertension, normotension, hypotension, or shock.5 Overall, 50% of patients present with hypertension. It is significantly more common for type B dissection to present with hypertension (70% of patients) as compared with type A dissection (36%). Hypotension or shock on presentation is significantly more common in type A dissection as compared with type B (25% and 5%, respectively).
Proximal dissection, involving the ascending aorta can result in one or more of the following clinical characteristics (Table 23.2):
Pericardial tamponade was found in 18.7% of patients with acute aortic dissection.18 It likely results from rupture of the aorta into the pericardial space. Tamponade has been found to correlate with presence of altered mental status and hypotension. Using logistic regression models, it was also found to be an independent predictor of outcome, even when controlling for baseline clinical findings.
TABLE 23.2 Complications of Type A Aortic Dissection
Complication
Incidence (%)
Tamponade
18.7
Acute aortic insufficiency
44
Myocardial ischemia
5
Neurologic manifestations
15
Various clinical complications can occur with type A aortic dissection. These, and their incidence among patients who present with acute type A aortic dissection are shown in the table.
Acute aortic insufficiency can be seen in up to 44% of patients presenting with type A dissection.5,19 There are several possible mechanisms of acute aortic insufficiency in the setting of acute dissection: (1) Incomplete coaptation of otherwise normal aortic valve owing to dilatation of the sinotubular junction, (2) abnormal leaflet coaptation owing to prolapse of the dissection flap into the valve, (3) extension of the dissection flap below the sinotubular junction resulting in prolapse of one or more of the aortic valve leaflets, (4) aortic leaflet prolapse related to a bicuspid aortic valve, and (5) degenerative aortic valve disease with leaflet thickening and abnormal coaptation.
Myocardial ischemia can complicate acute type A dissection by extension of the dissection flap into the coronary ostia. This can be seen in up to 5% of patients with type A dissection.5 The right coronary artery is most frequently involved.
Vocal cord paralysis from compression of the left recurrent laryngeal nerve has been reported.20
Neurologic manifestations, including altered mental status and focal neurological deficits or frank stroke, can complicate acute aortic dissection in up to 15% of patients.5,8 Neurologic complications can result from hypoperfusion secondary to shock or from direct extension of the dissection into the carotid vessels.
Descending thoracic aorta involvement can give rise to several distinct clinical characteristics including sudden onset of chest and back pain (86% of patients), abdominal pain, spinal cord ischemia (3% of patients), ischemic peripheral neuropathy (2% of patients), renal failure, shock, and lower extremity ischemia.5,21
Age and gender play a role in the clinical presentation of acute aortic dissection.8,22 Atherosclerosis, hypertension, and iatrogenic dissection are significantly more common in the older patient population, whereas Marfan syndrome-associated dissection is seen solely in the younger age group. Typical symptoms and signs of acute dissection (e.g., sudden onset of “aortic pain,” presence of aortic regurgitation murmur) are less common in the older (≥70 years) as compared with the younger (<70 years) patient population. Outcome and treatment of acute aortic dissection also differ between the age groups.
Women with acute aortic dissection are generally older and present later in the course of the disease.8 Pericardial or pleural effusion, tamponade, periaortic hematoma, altered mental status or coma, and hypotension are more frequent in women
and result in higher prevalence of in-hospital complications and mortality rate.
and result in higher prevalence of in-hospital complications and mortality rate.
TABLE 23.3 ECG Abnormalities in Aortic Dissection | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
DIAGNOSIS
The diagnosis of acute aortic dissection remains challenging despite significant improvements in imaging and diagnostic modalities. The overall occurrence of the disease is relatively rare; it is estimated that out of 1,000 patients who present to the emergency department with chest or back pain, only 3 patients are diagnosed with acute dissection.7 This is in contrast, for example, with acute coronary syndrome, which is probably 100 to 200 times more common. In one series,6 acute aortic dissection was the initial diagnosis in only 15% of patients who were ultimately diagnosed with acute dissection, demonstrating how challenging this diagnosis remains. Given the low incidence of aortic dissection and the overlap of presenting symptoms with other, more common disease processes, the diagnosis requires high level of suspicion and appropriate utilization of imaging studies in a timely manner.
Of the clinical characteristics, presence of aortic pain (abrupt onset of tearing or ripping pain), mediastinal widening on chest X-ray, and presence of pulse deficit or blood pressure differential were found to be very important for the diagnosis of acute dissection.23 In the absence of all three signs, the like-lihood of dissection was low (7%). In the presence of aortic pain or mediastinal widening the probability of dissection was intermediate (31% or 39%). In the presence of pulse deficit or blood pressure differential or any combination of the three characteristics, the probability of dissection was high, >83%.
Electrocardiographic (ECG) abnormalities can be seen in acute aortic dissection; however up to 30% of patients present with normal electrocardiogram.5 The ECG abnormalities that can be seen in acute dissection are summarized in Table 23.3.
Imaging studies are the key for the correct and timely diagnosis of acute aortic dissection (Figure 23.2). Currently available techniques include echocardiography (both transthoracic [TTE] and transesophageal [TEE]) contrast-enhanced computed tomography (CT), magnetic resonance imaging (MRI), and aortography.7,22,24,25,26




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree