Neonatal physiology
The distinctive and transitional features of the neonatal circulation may lead to cardiopulmonary abnormalities not only in newborn infants with cardiac malformations but also among those with pulmonary disease or other serious illnesses.
Understanding the anatomic and physiologic features of the transition from fetal to adult circulation aids the physician caring for critically ill neonates.
Normal fetal circulation
Normal fetal circulation differs from that of the postnatal state. In the fetus, the pulmonary and systemic circulations are parallel, rather than occurring in series as in the normal circulation. In the fetal circulation, both ventricles eject blood into the aorta and receive systemic venous return. The right ventricle ejects a greater volume than the left ventricle. Postnatally, the circulation differs because the ventricles and the circulation are in series. The right ventricle receives the systemic venous return and ejects it into the pulmonary artery. The pulmonary venous return passes through the left atrium and the left ventricle alone ejects blood into the aorta. Left ventricular and right ventricular outputs are equal. The transition from a parallel to a series circulation normally occurs soon after birth; however, in a distressed neonate, the parallel circulation may persist, delaying the evolution to series circulation.
The fetal circulation also has three distinctive anatomic structures: the placenta, the patent ductus arteriosus, and the patent foramen ovale. The blood returning to the fetus from the placenta enters the right atrium and flows predominantly from the right to the left atrium through the patent foramen ovale (Figure 8.1). This stream passes to the left ventricle and the ascending aorta, supplying the head with the proper level of oxygenated blood. The blood that leaves the head returns to the heart in the superior vena cava and flows principally into the right ventricle. Right ventricular output passes into the pulmonary artery, and the major portion (90%) flows through the ductus into the descending aorta, while a smaller amount (10%) flows into the lungs.
Figure 8.1 Central circulation in the fetus. Predominant flow from inferior vena cava is through the patent foramen ovale into the left atrium. The major portion of right ventricular flow is through the patent ductus arteriosus. AO, aorta; LA, left atrium; LV, left ventricle; PA, pulmonary artery; RA, right atrium; RV, right ventricle.
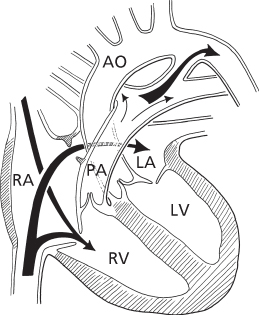
The major factor influencing the pattern and distribution of fetal blood flow is the relative vascular resistances of the pulmonary and systemic circuits. In contrast to the adult circulation, the pulmonary vascular resistance in the fetus is very elevated, and the systemic vascular resistance is low. Prenatally, the lungs are airless, and the pulmonary arterioles possess thick media and a narrowed lumen. These anatomic features of the pulmonary arterioles are accentuated by the relative hypoxic environment of the fetus, as hypoxia is a potent stimulus for pulmonary vasoconstriction. The systemic vascular resistance is unusually low, primarily because of the large flow through the placenta, which has low resistance. In the fetus, the pulmonary vascular resistance is perhaps five times greater than the systemic vascular resistance, the reverse of the adult circulation.
Because the systolic pressures in both ventricles and great vessels are identical, the distribution of blood flow depends upon the relative vascular resistances. As a result, a relatively small volume of blood flows through the lungs and a large volume passes through the ductus from right to left into the descending aorta. A considerable proportion (about 40%) of the combined ventricular output flows through the placenta.
The right-to-left shunt at the atrial level in the fetus depends in part upon the streaming effect caused by the position of the valve of the foramen ovale. This ridge tends to divert blood from the inferior vena cava into the left atrium through the defect. Since the atrial pressures are identical, the shunt also depends on the relative compliances of the ventricles. Approximately one-third of the total flow returning to the right atrium crosses the foramen ovale.
Transition to postnatal circulatory physiology
At birth, the distinctive features of fetal circulation and the vascular resistances are suddenly changed. A major reversal of resistance occurs because of the separation of the placenta and the onset of respiration. The loss of the placenta, which has acted essentially as an arteriovenous fistula, is associated with a doubling of systemic vascular resistance. The expansion of the lungs is associated with a sevenfold drop in pulmonary vascular resistance, principally from vasodilation of pulmonary arterioles secondary to an increase in inspired oxygen level to normal.
Coinciding with the fall in pulmonary vascular resistance, the volume of pulmonary blood flow increases and thus the volume of blood returning to the left atrium increases proportionately. The left atrial pressure rises, exceeds the right atrial pressure, and closes the foramen ovale functionally. In most infants for up to several months, a small left-to-right shunt occurs via the incompetent flap of the foramen ovale. Anatomically, the atrial septum ultimately seals in 75% of children and remains “probe-patent” in 25%.
The ductus narrows by muscular contraction within 24 hours of birth, although anatomic closure may take several days. The closure of the ductus is associated with a lowering of pulmonary arterial pressure to normal levels. When the ductus and foramen ovale close, the pulmonary blood flow equals systemic blood flow, and the circulations are in series.
In the neonatal period, the changes that occur in the ductus, foramen ovale, and pulmonary arterioles are reversible. The pulmonary arterioles and the ductus arteriosus are responsive to oxygen levels and acidosis. An increase in the vascular resistance occurs in conditions associated with hypoxia. Although minor changes occur at a PaO2 of 50 mmHg, large increases in pulmonary vascular resistance occur at PaO2 levels less than 25 mmHg. If acidosis coexists with hypoxia, the increase in pulmonary resistance is far greater than at comparable levels of PaO2 occurring at normal pH.
Persistent pulmonary hypertension of the newborn
Neonates with pulmonary parenchymal disease, such as respiratory distress syndrome, develop increased pulmonary vascular resistance and increased pulmonary arterial pressure because of hypoxia. If acidosis complicates the illness, the changes are even greater. This condition is often called persistent fetal circulation (PFC), or by the more physiologically descriptive term persistent pulmonary hypertension of the newborn (PPHN).
Because of the elevation of right ventricular systolic pressure, right atrial pressure increases, causing a right-to-left shunt at the foramen ovale. In a similar way, the ductus arteriosus of a neonate is also responsive to oxygen. With hypoxia, the ductus may reopen and, should the pulmonary resistance be simultaneously elevated, a right-to-left shunt could occur through the ductus arteriosus. Clinically, this is recognized by a lower PaO2 (or pulse oximetry saturation) in the legs than arms.
Thus, cyanosis in the neonate with pulmonary parenchymal disease can result from right-to-left shunting of blood, as well as from intrapulmonary shunting and diffusion defects. Administration of 100% oxygen improves both of these abnormalities, but often the improvement is not great enough to exclude cyanotic cardiac malformations. Administration of oxygen to cyanotic patients with a cardiac anomaly generally also lessens the degree of cyanosis. With the development of echocardiography, the ability to distinguish these has been greatly enhanced.
Cardiac disease in neonates
Most cardiac malformations cause no problems in utero or in the immediate postnatal state. However, during the transition to the normal circulatory pattern, particularly as the ductus arteriosus is closing and then closes, certain malformations become evident. These malformations have one of three circulatory patterns in which the ductus played an important role during fetal life, and as it closes postnatally the neonatal circulatory pattern is disrupted.
The three types of malformations dependent upon ductal blood flow following birth are as follows:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


