Chapter 3 Respiratory Mechanics
Introduction
This chapter describes the physical properties of the lungs and chest wall involved in the cyclic processes of ventilation supporting the metabolic needs of the body. The contribution of respiratory muscles to these processes is reviewed here; their function is described more fully in Chapter 6. Clinical measurements of some of these mechanical properties are an important part of pulmonary function testing, as discussed in Chapter 9.
Structure of the Thorax and Lungs
Thorax
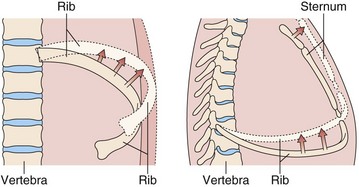
The bony thorax protects the lungs, heart, and great vessels but also allows the lungs to change volume from a minimum of 1.5 to 2.0 L to a maximum of 6 to 8 L. This large expansion is made possible by the articulation of the ribs with the spine and the sternum, the arrangement of the muscles, and the motion of the diaphragm. The ribs articulate with the transverse processes of the thoracic vertebrae and have flexible cartilaginous connections with the sternum. The ribs angle down, both from back to front and from midline to side, so that as they elevate, both the anteroposterior and the transverse dimensions of the thorax increase (Figure 3-1). The external intercostal muscles that angle down from posterior to anterior (Figure 3-2) are well situated to elevate the ribs. With deep inspiratory efforts, the first and second ribs are elevated and stabilized by the accessory muscles of respiration in the neck. If the upper extremities are fixed, the pectoralis muscles also can act to raise the ribs (e.g., leaning onto a chair back or against a wall when out of breath). Expiration normally is passive, driven by the elastic recoil of the lung, but can be assisted by the internal intercostal muscles. Forced expiration or a cough requires the abdominal muscles to force the diaphragm upward.
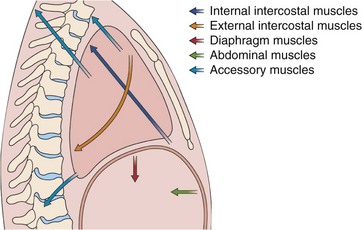
Figure 3-2 Action of the major respiratory muscle groups—intercostals, accessories, diaphragm, and abdominals.
The intercostal muscles are innervated from the thoracic spine at their own level, and the abdominal muscles are innervated from lower thoracic and lumbar level, but the diaphragm is served by the phrenic nerves, which originate at the cervical level (C3 to C5). Thus, the diaphragm remains functional in patients who have spinal injuries below the midcervical level. The long course of each phrenic nerve along the mediastinum, however, makes it vulnerable to both transient and permanent interruptions by disease, injury, or surgery. Occasionally, local irritation of a phrenic nerve leads to intractable singultus (i.e., hiccups). The respiratory muscles are more fully discussed in Chapter 6.
Airways
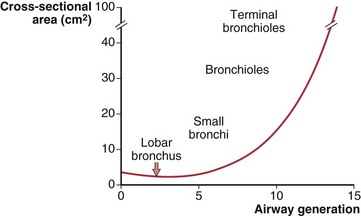
The number of airway generations required to reach the respiratory zone varies with pathway length, so that areas near the hilum may be reached in 15 generations, whereas those in the periphery may require 25 generations. Although the size of individual airways becomes smaller, the number of airways approximately doubles with each new generation, so that the total cross-sectional area of the combined air path increases. This is especially so in the smaller bronchi and bronchioles, where the “daughters” of each division are only slightly smaller than the “parent.” The rapidly increasing total cross-sectional area of small airways, shown diagrammatically in Figure 3-3, means that their contribution to airflow resistance in the lungs is small. Thus, diseases that affect these peripheral airways may be functionally silent until they reach an advanced state.
Respiratory Mechanics
Lung Volumes
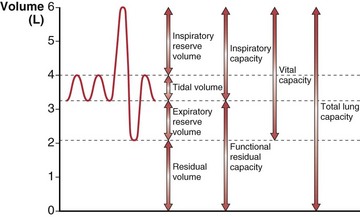
The total gas-containing capacity of the lungs can be divided into a series of “volumes,” as shown in Figure 3-4, which, in combination, give lung “capacities.” The largest amount of air that can be held in the lungs at full inspiration is the total lung capacity (TLC). After a complete forced exhalation, the lungs are not empty but contain a residual volume (RV). The difference between TLC and RV—that is, the greatest volume of air that can be inhaled or exhaled—is the vital capacity (VC). The vital capacity can be affected by factors that either limit expansion of the lung (restrictive processes) or limit lung emptying (airflow obstruction).
A normal breath has a tidal volume (VT) that is only a small portion of the vital capacity (approximately 10%), and even during strenuous exercise, VT increases to only 50% to 60% of VC. Increases in VT occur by extending into the inspiratory reserve and expiratory reserve volumes as shown in Figure 3-4. At the end of a relaxed tidal exhalation, the lungs and chest wall return to a resting position, which normally is approximately 50% of TLC. The volume contained in the lungs at this end-tidal position is the functional residual capacity (FRC), and the volume that can be inhaled from this point is the inspiratory capacity (IC).
The Lung–Chest Wall System
Volumes of Elastic Structures
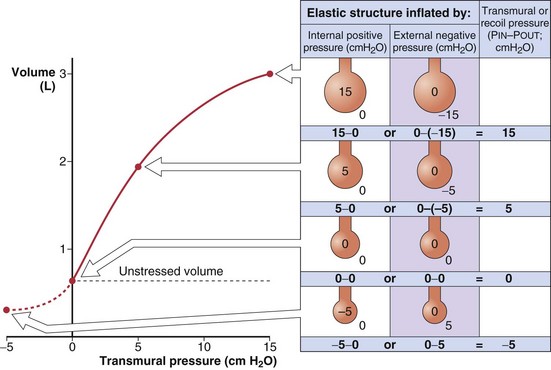
The recoil tendency of a spring can be expressed in terms of its unstressed or resting length and its length-tension relationship. Similarly, for expandable volumetric structures, the relevant properties are the unstressed volume and the relationship between volume and the transmural pressure required to achieve that volume (Figure 3-5). By convention, transmural pressures are expressed as the difference between the pressure inside and the pressure outside the structure (Pin – Pout). It is convenient to think of this as the distending pressure required to achieve a certain volume. In addition, this distending pressure also represents the recoil pressure, or the tendency of the structure to return to its unstressed volume (where transmural pressure is zero). A positive recoil pressure indicates a tendency to become smaller. A structure distorted to a volume below its unstressed volume has a negative recoil pressure, which indicates its tendency to become larger.
Elastic Properties of the Lung
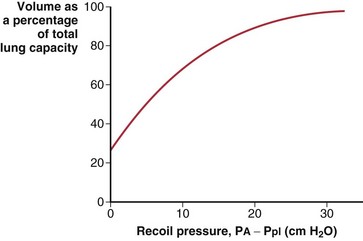
The lungs are elastic structures with a tendency to recoil to a small “unstressed volume” (usually slightly less than RV). To maintain any lung volume larger than this unstressed volume requires a force that distends the lungs; this force is the difference between the alveolar pressure (PA) and the pressure surrounding the lungs, the intrapleural pressure (Ppl). The elastic properties of the lungs and their tendency to recoil are represented by a plot of the relationship between lung volume and transmural pressure (Figure 3-6). Such graphs apply to an excised lung being inflated by a pump, an in vivo lung inflated by a ventilator, or the more physiologic normal lung inflated by expanding the chest (to create a more negative pleural pressure). In each case, the curve of volume versus the transpulmonary pressure difference (PA − Ppl) is the same.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree