Chapter 22 Pulmonary Host Defenses
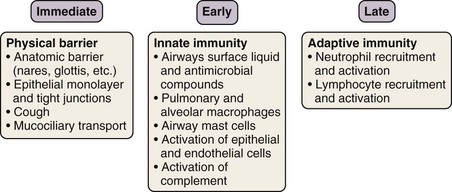
The epithelial surface of the lung is continuously exposed to a variety of potentially pathogenic microorganisms, allergens, particulate pollutants, and other noxious agents. An intricate defense system has evolved over time to protect the lungs from these potentially harmful entities while preserving homeostasis and lung function (Figure 22-1). This system of defense mechanisms has two components: an innate (nonspecific) response and an adaptive or acquired (specific) response.
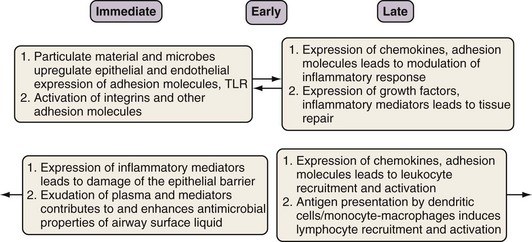
The innate immune response is evolutionarily conserved to provide immediate (occurring over seconds to minutes) host defense in a broad, nonspecific manner. Only vertebrates have an additional, adaptive immune system, which is directed at specific pathogens or molecules. Although the two systems work in concert to protect the host, each has several distinctive features. With rare exceptions, the innate component of the immune system depends on proteins and signaling pathways that exist in a fully functional form, does not require priming, and is not strengthened with subsequent exposures. By contrast, the adaptive immune response requires additional time (days to weeks) (see Figures 22-2 and 22-3) to ramp up to full capacity, is specific to the pathogen (and even to specific molecular determinants of the pathogen), and has memory to provide for stronger responses with subsequent attacks (“anamnestic response”).
Structural Defenses
With a surface area in adults of 70 m2, which comes into contact with roughly 10,000 L of air a day, the lung is confronted with constant threats from microbes. In addition to inhaled pathogens, high bacterial concentrations are present in oropharyngeal secretions (108/mL), and aspiration of these secretions also may pose a serious risk for invasive infection (aspiration pneumonia). Historically, the lung has been considered to be a sterile environment; recent developments in molecular analysis have provided evidence for the presence of a microbial flora of considerable diversity in the lung, which moreover is altered in disease states. Accordingly, the respiratory tract has developed a series of structural barriers that are designed both to minimize the number of pathogenic microbes entering the lungs and to hasten their clearance before an infection can be established (Table 22-1).
Table 22-1 Structural Defenses of the Airway
| Structure | Functions |
|---|---|
| Nose | |
| Glottis | |
| Mucociliary escalator | |
| Epithelium |
Particle size is an important factor determining the degree of penetration into the airways (Table 22-2). Very large particles are filtered by vibrissae (nasal hairs). Particles approximately 30 µm in diameter are removed in the nasal passages, where turbulent airflow results in prolonged air-mucosa contact, with subsequent particle impaction. Most particles between 10 and 30 µm in diameter also will be deposited on the turbinates and nasal septum, carina, or within the larger bronchi. The branching nature of the airways provides two additional mechanisms of protection: (1) the secondary, tertiary, and quaternary carinae force particles to embed in the airway mucosa, thereby preventing further penetration into the lung, and (2) reduced airflow with increased airway branching allows gravity to sediment most particles larger than 2 µm. Particles less than 0.2 to 0.5 µm across tend to stay suspended as aerosols and are exhaled. Much smaller particles (less than 0.1 µm) may be deposited as a result of brownian motion (bombardment with gas molecules).
Table 22-2 Effect of Particle Size on Penetration into Airways
| Particle Size | Fate |
|---|---|
| >>>30 µm | Filtered by vibrissae |
| >30 µm | Nasopharyngeal impaction |
| 10 to 30 µm | Nasopharyngeal, tracheal, and large bronchial impaction |
| 2 to 10 µm | Sedimentation in airways |
| 0.2 to 2 µm | Reach alveoli |
| 0.2 to 0.5 µm | Exhaled |
| <0.2 µm | Exhaled or deposited (brownian motion) |
Cough/Sneeze
Mechanical or chemical stimulation of receptors in the nose, larynx, or trachea or elsewhere in the respiratory tree may produce bronchoconstriction to prevent deeper penetration of irritants and also may trigger the cough or sneeze reflex to expel particles deposited in the airways (Table 22-3). The cough reflex aids mucociliary transport to remove trapped particles. Mucus usually is conveyed to the carina by the cilia and then expelled by coughing from this location. Disruption of the cough reflex (e.g., in smokers or patients with vocal cord palsy or stroke) results in a predisposition to pneumonias.
Table 22-3 Phases of Cough/Sneeze
| Component | Event(s) |
|---|---|
| 1. Inspiratory phase | Deep inspiration, usually 1 to 2 times tidal volume |
| 2. Compression phase | Begins with closure of the glottis and contraction of respiratory muscles, resulting in the generation of high intrathoracic pressure (up to 100 to 200 cm H2O in adults) |
| 3. Expressive phase | Glottal opening, with airflow at rates as high as 25,000 cm/sec (partly helped by compression of airway cross section) |
| 4. Relaxation phase | Relaxation of respiratory muscles with temporary bronchodilatation |
Respiratory Epithelium
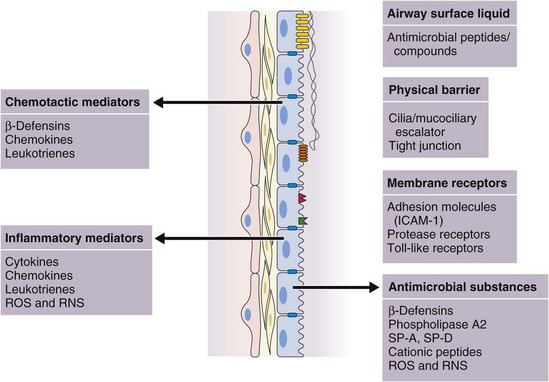
In addition to this important physical barrier function, epithelial cells, including ciliated cells, goblet cells, serous cells, basal cells, and Clara cells, are integral to the normal function of the mucociliary escalator, the production of a variety of antimicrobial molecules, and the initiation and regulation of inflammatory responses (see Figure 22-4).
Mucociliary Escalator
The airway epithelium is lined from the trachea to the respiratory bronchioles by the airway surface liquid (ASL), a 5- to 25-µm-thick surface film the primary function of which is to trap and facilitate the physical removal of foreign particles, as well as to provide an environment conducive to the activity of antimicrobial molecules (Table 22-4).
Table 22-4 Properties of the Mucociliary Escalator
| Structure | Function(s) | Dysfunction in Disease |
|---|---|---|
ASL is a critical component of the mucociliary escalator and is the result of secretion by glands and serous cells and of plasma transudation (Table 22-5). The inner low-viscosity periciliary sol facilitates the coordinated beating action of the cilia that propels the outer viscous mucous blanket toward the glottis, thereby facilitating the removal of trapped pathogens or particles by expectoration or ingestion. The viscous mucus is composed of mucopolysaccharides, produced predominantly by submucous glands in the larger airways, with increasing contributions from goblet cells and Clara cells with successively larger airway generations.
Table 22-5 Properties of Airway Surface Liquid (ASL)
| Composition | Function | Dysfunction in Disease |
|---|---|---|
| Secretions from glands and goblet cells Plasma transudation | Antimicrobial properties due to low pH and secreted antimicrobial compounds | Cystic fibrosis Asthma COPD |
COPD, chronic obstructive pulmonary disease.
Specific Cell Responses
Diverse cell populations contribute to the host defense system of the lung. In addition to the cells of the respiratory epithelium, these include other structural cells of the lung, the pulmonary vascular endothelium and fibroblasts, resident leukocytes, and, at later stages of immune responses, recruited immune cells (Table 22-6), as reviewed next.
Table 22-6 Cells Mediating Pulmonary Host Defense and the Inflammatory Response
| Timing | Immune Cells | Nonimmune Cells |
|---|---|---|
| Early phase | ||
| Late phase |
Epithelial Cells
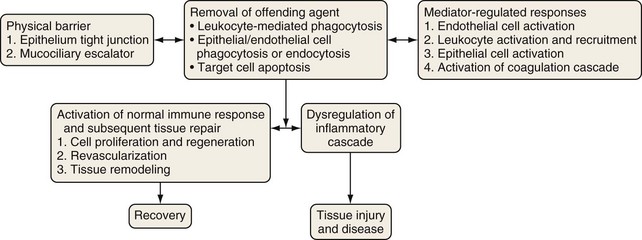
The contribution of the respiratory epithelium is not limited to its roles as a structural barrier and facilitator of mucociliary clearance (Box 22-1). Respiratory epithelial cells actively participate in the regulation of inflammation and are capable of mounting an immune response by internalization of organisms and secretion of cytotoxic and antimicrobial peptides. Inhaled microbial pathogens including bacteria and viruses and other antigens can trigger activation of pathogen recognition receptors (such as Toll-like receptors [TLRs], discussed later in some detail under “Innate Immune Receptors”) expressed by epithelial cells. Epithelial cells are induced by bacterial components, such as LPS, and by cytokines such as tumor necrosis factor (TNF)-α and interleukin-1β (IL-1β) to express various gene products (by the NFκB signaling pathway, discussed later on) that modulate the inflammatory response (Figure 22-4). Such inducers include the following:
• Cytokines such as TNF-α, IL-1β, and thymic stromal lymphopoietin (TSLP)
• Chemokines that include macrophage inflammatory protein (MIP)-2, CXC chemokines, monocyte chemoattractant protein (MCP)-1, IL-7, IL-8, IL-15
• Nitric oxide (NO) and reactive nitrogen species (ONOO−)
• Adhesion molecules such as β-integrins and intercellular adhesion molecule-1 (ICAM-1)
• TLRs such as TLR-2 and TLR-4
• TNF-α receptors, TNFR1 and TNFR2
• Growth factor receptors such as epidermal growth factor receptor (EGFR) and platelet-derived growth factor receptor (PDGFR)