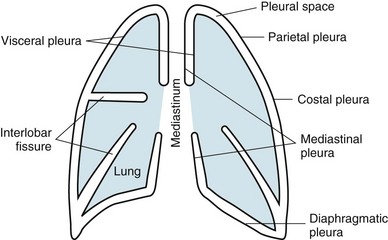
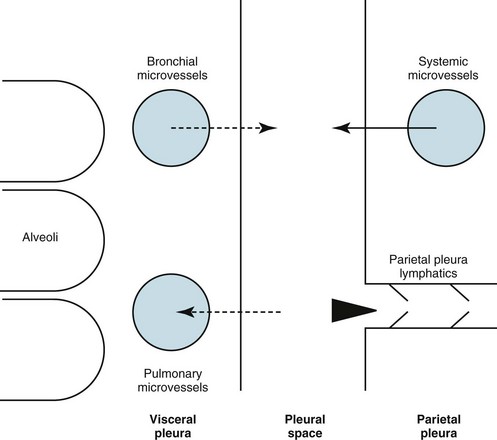
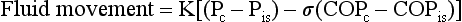15 This chapter discusses the anatomy of the pleura, followed by a presentation of several physiologic principles of fluid formation and absorption by the pleura and a discussion of two types of abnormalities that affect the pleura: liquid in the pleural space (pleural effusion) and air in the pleural space (pneumothorax). A comprehensive treatment of all the disorders that affect the pleura is beyond the scope of this text. Rather, this chapter aims to cover the major categories and give the reader an understanding of how different factors interact in producing pleural disease. The primary malignancy of the pleura, mesothelioma, is discussed in Chapter 21, which deals with neoplastic disease of the thorax. The term pleura refers to the thin lining layer on the outer surface of the lung (visceral pleura), the corresponding lining layer on the inner surface of the chest wall (parietal pleura), and the space between them (pleural space) (Fig. 15-1). Because the visceral and parietal pleural surfaces normally touch each other, the space between them is usually only a potential space. It contains a thin layer of serous fluid coating the apposing surfaces. When air or a larger amount of fluid accumulates in the pleural space, the visceral and parietal pleural surfaces are separated, and the space between the lung and the chest wall becomes more apparent. The pleura lines not only the surfaces of the lung in direct contact with the chest wall but also the diaphragmatic and mediastinal borders of the lung. These surfaces are called the diaphragmatic and mediastinal pleura, respectively (see Fig. 15-1). Visceral pleura also separates the lobes of the lung from each other; therefore, the major and minor fissures are defined by two apposing visceral pleural surfaces. The pleural space normally contains only a small quantity of liquid (≈10 mL), which lubricates the apposing surfaces of the visceral and parietal pleurae. According to the current concept of pleural fluid formation and resorption, formation of fluid is ongoing primarily from the parietal pleural surface, and fluid is resorbed through the stomata into the lymphatic channels of the parietal pleura (Fig. 15-2). The normal rates of formation and resorption of fluid, which must be equal if the quantity of fluid within the pleural space is not changing, are believed to be approximately 15 to 20 mL/day. where K = filtration coefficient (a function of the permeability of the pleural surface), P = hydrostatic pressure, COP = colloid osmotic pressure, σ = measure of capillary permeability to protein (called the reflection coefficient), and the subscripts c and is refer to the capillary and pericapillary interstitial space, respectively. In this case, the pericapillary interstitial space is essentially the pleural space; therefore, Pis and COPis refer to intrapleural pressure and the colloid osmotic pressure of pleural fluid, respectively. The intrapleural pressure—that is, the hydrostatic pressure within the pleural space—is negative, reflecting the outward elastic recoil of the chest wall and the inward elastic recoil of the lung. The numerous causes of pleural fluid accumulation are best divided into transudative and exudative categories (Table 15-1). This distinction is generally easy to make and is most important in guiding the physician along the best route for further evaluation. Transudative fluid usually implies that the pathologic process does not primarily involve the pleural surfaces, whereas exudative fluid often suggests that the pleura is affected by the disease process causing the effusion. Table 15-1 MAJOR CAUSES OF PLEURAL EFFUSION TRANSUDATE Increased hydrostatic pressure; “overflow” of liquid from the lung interstitium Decreased plasma oncotic pressure Movement of transudative ascitic fluid through the diaphragm EXUDATE
Pleural Disease
Anatomy
Physiology
Pleural Effusion
Etiology of Pleural Effusion
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Pleural Disease