To determine whether non-O blood group is an independent risk factor for coronary artery disease (CAD), we performed a meta-analysis of contemporary studies reporting adjusted relative risk estimates using multivariable logistic regression and multivariable Cox proportional hazards regression. MEDLINE and EMBASE were searched from January 2001 to March 2015 using Web-based search engines (PubMed and OVID). Search terms included blood group/type and coronary (artery/heart) disease, ischemic/ischaemic heart disease, acute coronary syndrome, myocardial infarction, or angina. Eligible were case-control, cross-sectional, or cohort studies reporting adjusted odds ratios (ORs) and hazard ratios (HRs) for CAD in subjects with non-O versus O blood group using multivariable logistic regression/multivariable Cox proportional hazards regression. Study-specific estimates were combined using inverse variance-weighted averages of logarithmic ORs/HRs in the random-effects model. Of 117 potentially relevant articles screened initially, 12 relative risk estimates in 10 eligible studies were identified and included. Pooled analysis of all the 12 ORs/HRs demonstrated that non-O blood group was associated with a statistically significant 14% increase in CAD incidence relative to O blood group (OR/HR 1.14, 95% confidence interval 1.04 to 1.25, p = 0.006). There was no evidence of significant publication bias. Combining 8 studies reporting data regarding (acute) myocardial infarction also generated a statistically significant result unfavoring non-O blood group (OR/HR 1.16, 95% confidence interval 1.02 to 1.31, p = 0.02). In conclusion, we found that based on a meta-analysis of 10 studies enrolling a total of 174,945 participants, non-O blood group appears to be an independent risk factor for CAD and myocardial infarction.
Subjects with non-O blood group are suggested to have a higher risk of developing thrombotic episodes of venous or arterial origin. According to previous meta-analyses, although non-O blood group is a candidate to be one of the most important genetic risk factors for venous thrombosis, the non-O risk for myocardial infarction (MI) is restricted to retrospective studies with no overall effect observed in prospective cohorts. The results of a recent meta-analysis confirm a weak association between non-O blood group and vascular arterial thrombosis, in particular myocardial ischemia. Investigators of these previous meta-analyses abstracted “crude” relative risk estimates, generated simply using event (non-O blood group) rates in both the case and control groups, from each individual study and then combined them. In such analyses (“unadjusted” univariable analyses and meta-analyses of their results), however, it can be never determined whether non-O blood group is an “independent” risk factor for coronary artery disease (CAD). To identify independent risk factors, multivariable logistic regression (MLR) and multivariable Cox proportional hazards regression (MCPHR) must be used, generating an “adjusted” odds ratio (OR) and hazard ratio (HR), respectively. To determine whether non-O blood group is an independent risk factor for CAD, hence, we performed a meta-analysis of contemporary studies reporting adjusted relative risk estimates using MLR/MCPHR.
Methods
All eligible studies were identified using a 2-level search strategy. First, databases including MEDLINE and EMBASE were searched from January 2001 to March 2015 using Web-based search engines (PubMed and OVID). Second, relevant studies were identified through a manual search of secondary sources including references of initially identified articles and a search of reviews and commentaries. Search terms included blood group/type and coronary (artery/heart) disease, ischemic/ischaemic heart disease, acute coronary syndrome, myocardial infarction, or angina.
Studies considered for inclusion met the following criteria: the design was a case-control, cross-sectional, or cohort study; the study population was subjects with or without CAD; and main outcomes included adjusted relative risk estimates for CAD in subjects with non-O versus O blood group using MLR/MCPHR. We selected contemporary (published in the 21st century [posterior to 2001]) studies.
Data regarding an adjusted OR/HR for CAD in subjects with non-O versus O blood group were abstracted (as available) from each individual study. In a number of studies, a statistically nonsignificant, adjusted OR/HR was unavailable. If the unavailable, statistically nonsignificant, adjusted ORs/HRs for non-O versus O blood group were ignored and not included in a meta-analysis, the pooled result would be biased unfavoring non-O blood group. In such a case, thus, we extracted a statistically nonsignificant, unadjusted OR/HR (if available), instead of the unavailable, statistically nonsignificant, adjusted OR/HR.
Study-specific estimates were combined using inverse variance-weighted averages of logarithmic ORs/HRs in the random-effects model. Sensitivity analyses were performed to assess the contribution of each study to the pooled estimate by excluding individual studies one at a time and recalculating the pooled OR/HR estimates for the remaining studies. To assess the impact of study design on the pooled estimate, the effects of non-O blood group were explored separately in case-control, cross-sectional, and cohort studies. Publication bias was assessed graphically using a funnel plot and mathematically using an adjusted rank-correlation and linear regression test. All analyses were conducted using Review Manager, version 5.3 (Nordic Cochrane Center, Copenhagen, Denmark) and Comprehensive Meta-Analysis, version 2 (Biostat, Englewood, New Jersey).
Results
Of 117 potentially relevant articles screened initially, 14 relative risk estimates in 10 eligible studies ( Table 1 ) were identified and included. In total, our meta-analysis included data on 174,945 participants with 21,570 cases (patients with CAD). Despite noted heterogeneity in the definition of CAD among the studies, we judged to merit inclusion of all studies in the quantitative meta-analysis. Pooled analysis of all the 12 ORs/HRs demonstrated that non-O blood group was associated with a statistically significant 14% increase in CAD incidence relative to O blood group (OR/HR 1.14, 95% confidence interval [CI] 1.04 to 1.25, p = 0.006; Figure 1 ).
| Case-control study | ||||||||
|---|---|---|---|---|---|---|---|---|
| Study | Definition | Number | RRE for non-O versus O | |||||
| Case | Control | Case | Control | RRE | PE | LLCI | ULCI | |
| Biswas 2013 | Typical angina and evidence of ischemia or infarction after ElCG study, TMT, stress EcCG and EcCG study | Spouses, neighbors, and people from same work place of the patients, with the same sociocultural background, in whom the clinical history, the objective search for signals of CAD, and the ElCG as well as EcCG examinations did not suggest the presence of CAD | 250 | 250 | OR for CAD | 0.54 | 0.32 | 0.90 |
| Franchini 2013 | CAD | Healthy general population (blood donors) | 1879 | 4272 | OR for CAD | 1.50 | 1.06 | 2.11 |
| Nydegger 2003 | Survivors of an AMI that had occurred at least 2 months before inclusion | Healthy controls, without a history of thromboembolic events or tendency to bleed (frequency-matched to the cases by age [SD, 5 years] and sex) | 177 | 89 | OR for AMI | 0.74 | 0.43 | 1.26 |
| Tanis 2006 | Women with a first AMI between 18 and 49 years of age | Women contacted by random-digit dialing, stratified for age, index year of MI, and area of residence | 200 | 626 | OR for AMI | 1.59 | 1.10 | 2.30 |
| Cross-sectional study | ||||||||
|---|---|---|---|---|---|---|---|---|
| Study | Participant | Diagnosis | Case | Participant | RRE for non-O versus O | |||
| Chen 2014 | Patients undergoing diagnostic or interventional CAG | Significant angiographically documented CAD | 4377 | 6242 | OR for CAD | 1.24 | 1.10 | 1.39 |
| Prevalent or incident MI | 1974 | OR for MI | 1.22 | 1.09 | 1.37 | |||
| Gong 2014 | Patients undergoing selective CAG for being suspected or known coronary atherosclerosis | Significant angiographically documented CAD as having >50% diameters stenosis in ≥1 major coronary artery | 3343 | 3806 | OR for CAD | 1.49 | 1.20 | 1.84 |
| MI according to the World Health Organization criteria | 980 | OR for MI | 1.24 | 1.04 | 1.47 | |||
| Lee 2012 | Men younger than 45 years and women younger than 55 years undergoing CAG | CAD defined as >50% stenosis in ≥1 major coronary branch on CAG | 136 | 265 | OR for CAD | 0.94 | 0.42 | 2.08 |
| AMI diagnosed as per the universal definition | 73 | OR for AMI | 0.52 | 0.19 | 1.45 | |||
| Reilly 2011 | Patients of European ancestry | Angiographic CAD | 933 | 1401 | OR for CAD | 1.13 | 0.90 | 1.43 |
| Angiographic MI | 470 | OR for MI | 1.44 | 1.10 | 1.90 | |||
| Cohort study | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Study | Participant | Event | Follow-up | Event | Participant | RRE for non-O versus O | |||
| CCHS 2013 | Adult Danish general population | MI defined as ICD-8 code 410 and ICD-10 codes I21–I22 | From 1977 through 2010 | 1045 | 9041 | HR for MI | 1.10 | 0.95 | 1.27 |
| CGPS 2013 | 1663 | 56,946 | 1.00 | 0.90 | 1.10 | ||||
| HPFS 2012 | Men aged 40 to 75 years | CAD (nonfatal MI or fatal CAD) through a biennial questionnaire regarding medical conditions | 20 years | 2015 | 27,428 | HR for CAD | 1.09 | 0.99 | 1.20 |
| NHS 2012 | Female nurses aged 30 to 55 years | 26 years | 2055 | 62,073 | 1.12 | 1.02 | 1.24 | ||
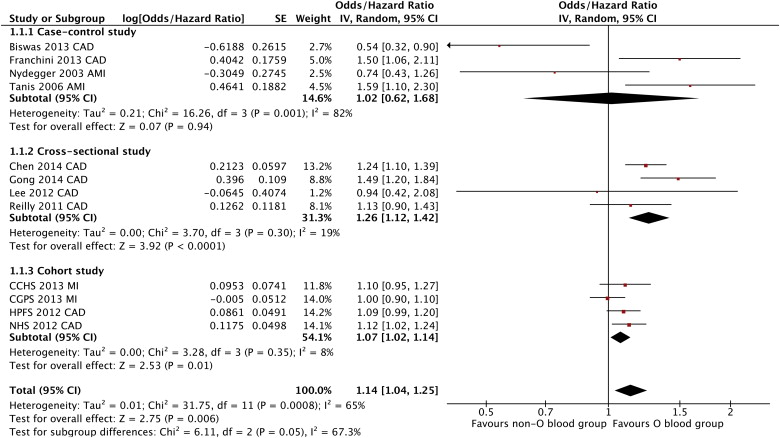
To assess the impact of qualitative heterogeneity in study design and participant selection on the pooled effect estimate, we performed several sensitivity analyses. Exclusion of any single study from the analysis did not substantively alter the overall result of our analysis ( Figure 2 ). When data from case-control, cross-sectional, and cohort studies were pooled separately, non-O relative to O blood group was associated with a statistically significant increase in CAD incidence in both cross-sectional (representing 11,714 participants) and cohort studies (representing 155,488 participants) but no increase in CAD incidence in case-control studies (representing 7,743 participants; OR/HR 1.02, 95% CI 0.62 to 1.68, p = 0.94; Figure 1 ). Additionally, combining 8 studies reporting data regarding (acute) MI generated a statistically significant result unfavoring non-O blood group (OR/HR 1.16, 95% CI 1.02 to 1.31, p = 0.02; Figure 3 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


