Category
Systolic
Diastolic
Optimal
<120
And
<80
Normal
120–129
And/or
80–84
High normal
130–139
And/or
85–89
Grade 1 hypertension
140–159
And/or
90–99
Grade 2 hypertension
160–179
And/or
100–109
Grade 3 hypertension
≥180
And/or
≥110
Isolated systolic hypertension
≥140
And
<90
This classification is very important because it heightens awareness of both risk and opportunities for prevention, adopting therapeutic management based on lifestyle changes, and adding incremental drug therapy if necessary.
Hypertension has both modifiable and nonmodifiable risk factors. Gender and genetic heritage are nonmodifiable factors, although the latter is uncertain. As we discussed in a recent review [3], epigenetic inheritance plays a key role in determining the activation of the genome and the axis of stress. This type of information is a stable genetic trace, transmitted from generation to generation, but can gradually be changed and modulated by lifestyle and the surrounding environment (for details, see the section “Allostatic load and the building of cardiovascular diseases” in the aforementioned review, part B).
Systolic blood pressure rises throughout the adult age range, whereas diastolic blood pressure peaks at about age 60 years in men and 70 years in women and falls gradually thereafter [1]. Even if both systolic and diastolic blood pressures are independently predictive of stroke and coronary mortality, in the elderly, a wide pulse pressure (systolic blood pressure minus diastolic blood pressure) has been shown in some observational studies to be a better predictor of adverse cardiovascular outcomes than either systolic or diastolic pressures individually [1]. Thus, hypertension has always been considered a condition of aging: more than 65% of persons 65 years old or older are hypertensive, but, surprisingly, a person who is normotensive at age 55 years still has a 90% lifetime risk of developing hypertension [4]. This seems to strongly suggest that hypertension might be the result of an integrated and multifaceted individual adaptation to the environment and dramatically emphasizes the importance of environmental factors together with individual psychosocial and emotional factors in dealing with this issue [3].
Rather than focusing on chronological aging, the emphasis should be on physiological aging, which can be changed.
As we will see, lifestyle modifications demonstrably reduce BP: losing weight and certain diets, sodium intake reduction, moderate physical exercise, alcohol consumption reduction, smoking cessation, and stress and pain management.
This is very important because there is a considerable gap between what has been shown in pharmaceutical trials and what happens in daily clinical practice in terms of achievable BP reductions. Although pharmaceutical therapy has been demonstrated to be effective in reducing heart attacks and stroke, outside of clinical trials, only approximately one-third of patients achieve optimal BP control using drug therapy [2]. This efficacy gap in hypertension treatment represents an ideal opportunity to codevelop with a patient a customized action plan that addresses logical options for diet, exercise, supplementation, smoking cessation, and mind–body skills development [5]. Additional insights may also come from both Ayurvedic and traditional East Asian medicine.
In this chapter, several examples regarding the anti-hypertensive effectiveness of each individual factor previously listed are reviewed. Each of them can then be associated with obtaining a synergistic effect [5]. This can be very important because a reduction of just 5 mmHg in systolic BP is associated with a 7% reduction in all-cause mortality [6].
Hypertension is often dubbed the “silent killer” because it is frequently asymptomatic; the pressure value remains as an abstract number devoid of real significance for the patient. If no pain is felt, no disease is present. Thus, it is problematic to arrange a treatment for a condition that is not perceived by the subject and, in addition, a chronic therapy, of long duration.
It is essential that the doctor can find the time to go on a therapeutic journey with the patient, picking up and respecting the patient’s beliefs, meanings, and interpretations of personal experience of hypertension and all that accompanies it.
Another key aspect in the management of the hypertensive patient is awareness of the problem and the fact that the condition persists even at home, in everyday life, and that it is not just a clinical quirk. Home BP monitoring with records in a diary or 24-h BP digital monitoring are simple means of raising awareness and compliance [1]. The data collected from the patient can be employed by the physician to educate the patient to take care of his health. They can be used to fill with him his individual cardiovascular risk score monitoring how it can improve over time (to this end, an electronic calculator can be used, such as: http://my.clevelandclinic.org/services/heart/prevention/risk-factors/Heart_Center_Risk_Tool).
More and more people ask not strictly pharmacological and symptomatic therapies for their health problems [7]. They seek integrative clinicians who can counsel from an evidence base on the logical options available to them (Fig. 15.1). Prehypertension is an early warning to modify lifestyle and probably is the ideal scenario to get the best results from an integrative approach. In addition, the change of lifestyle is a fundamental step, which is boosted at any degree of hypertension (Table 15.2). The physician should try to increase his/her knowledge about nonpharmacological interventions that can be adopted in the treatment of hypertension, moving beyond the basic and often vague recommendations on lifestyle modifications that can be found in guidelines [2] or traditional books [1].
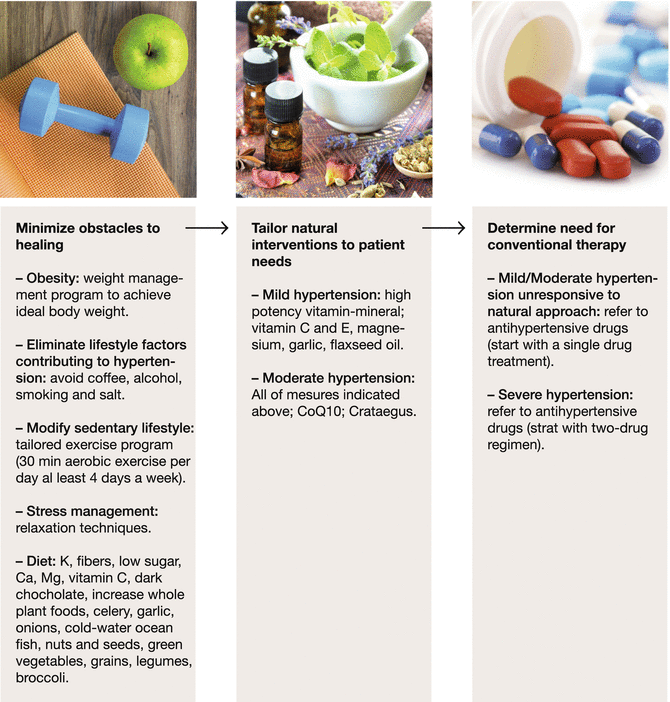

Fig. 15.1
In summary: staged integrative approach to hypertension. Eat food, not too much and mostly plants. Exercise 30 min per day for at least 4 days per week. Limit alcohol. Don’t smoke. Breathe: incorporate mind–body practices into your daily routine. Build an organized system of regular follow-up and review with self-monitoring and appointment reminders
Table 15.2
Initiation of antihypertensive treatment
Other risk factors, OD, or disease | Normal SBP 120–129 or DBP 80–84 | High normal SBP 130–139 or DBP 85–89 | Grade 1 HT SBP 140–159 or DBP 90–99 | Grade 2 HT SBP 160–179 or DBP 100–109 | Grade 3 HT SBP ≥180 or DBP ≥110 |
|---|---|---|---|---|---|
No other risk factors | No BP intervention | No BP intervention | Lifestyle changes for several months then drug treatment if BP uncontrolled | Lifestyle changes for several weeks then drug treatment if BP uncontrolled | Lifestyle changes+immediate drug treatment |
1–2 risk factors | Lifestyle changes | Lifestyle changes | Lifestyle changes for several weeks then drug treatment if BP uncontrolled | Lifestyle changes for several weeks then drug treatment if BP uncontrolled | Lifestyle changes +immediate drug treatment |
3 or more risk factors, MS, or OD | Lifestyle changes | Lifestyle changes and consider drug treatment | Lifestyle changes+ drug treatment | Lifestyle changes+ drug treatment | Lifestyle changes+immediate drug treatment |
Diabetes | Lifestyle changes | ||||
Established CV or renal disease | Lifestyle changes+immediate drug treatment | Lifestyle changes+immediate drug treatment | Lifestyle changes+immediate drug treatment | Lifestyle changes +immediate drug treatment | Lifestyle changes +immediate drug treatment |
15.2 The Role of Oxidative Stress
Oxidative stress (an imbalance of harmful oxygen species and the anti-oxidant defense mechanism) represents an etiological factor of human hypertension [8–10], hypertensive patients have a higher level of oxygen free radicals [11] and an impaired anti-oxidant defense mechanism [12]. This, together with an imbalance in vasodilators (nitric oxide) and vasoconstrictors (angiotensin), contributes to the initiation and perpetuation of hypertension [13]. This is particularly important because oxidative stress depends on external factors (alimentation, pollution, etc.), but also on inner factors such as individual psychological profile [3, 14, 15]. Thus, it is of paramount importance to act at both these levels.
15.3 Lifestyle Modification
15.3.1 Smoking Cessation
Smoking (tobacco addiction) is the most significant of the modifiable cardiovascular risk factors [16]. At least 1 billion adults worldwide are smokers and at least 700 million children are passive smokers at home [17]. Many people mistakenly describe their tobacco addiction as a “habit” or “behavioral choice,” and the onset of this state quickly follows the acquisition of an ability to inhale cigarette smoke, leading to a transformation of neurophysiological function and nicotine-receptor density [16]. To maintain the personal “comfort,” the patient becomes addicted to nicotine [3]. Smokers inhale thousands of other chemicals, many of which play critical roles in the initiation and accentuation of atherosclerosis by influencing vasomotor activity, vascular dysfunction, oxidation of lipids, atheroma development, and thrombosis [1].
Smoking cessation, of course, should be part of every comprehensive lifestyle modification plan. The use of cigarettes is reported to determine a 4-mmHg increase in systolic blood pressure and a 3-mmHg increase in diastolic blood pressure [18], probably by increasing the resting heart rate [19]. Furthermore, hypertensive patients who smoke are at an additional increased risk for cardiovascular events (ischemic stroke and hemorrhagic stroke) compared with those who do not smoke, and this risk correlates directly with the number of cigarettes smoked [20, 21].
15.3.2 Diet
A diet effective in the treatment of hypertension must include vegetables, fruits, and low-fat dairy products and should be low in saturated fat and refined grains/carbohydrates, not high in sodium, but rich in potassium, magnesium, calcium, and fiber.
Two very well-studied diets for hypertension prevention and control are the Mediterranean [14] and DASH (Dietary Approach to Stop Hypertension) [5] diets.
15.3.2.1 Mediterranean Diet
The Mediterranean diet’s antihypertensive effect was evaluated in the SUN study, a prospective cohort study conducted from 1999 to 2005 in 9,408 men and women [22]. The study documented a mean systolic BP reduction of 3.1 mmHg (95% confidence interval [CI] 5.4 to −0.8) and mean diastolic BP decrease of 1.9 mmHg (95% CI 3.6 to −0.1) [22].
The benefits of the Mediterranean diet are beyond the anti-hypertensive effect taken in isolation. Foods that we introduce into the diet are not only a source of calories, but are important modulators of the nervous, endocrine, and immune systems [14]. Thus, beyond hypertension, it has been demonstrated that the Mediterranean diet has favorable effects on lipoprotein levels, endothelium vasodilation, insulin resistance, metabolic syndrome, anti-oxidant capacity, myocardial and cardiovascular mortality, and cancer incidence in obese patients and in those with previous myocardial infarction [23, 24].
Additionally, adherence to the Mediterranean diet was demonstrated to be associated with approximately a 20% reduction in all-cause mortality in both men and women [25]. This percentage doubles in the elderly population [26].
One of the main components of the Mediterranean diet is olive oil, rich in healthy polyphenols and monounsaturated fatty acids (oleic acid) with an anti-hypertensive effect [27, 28]. In addition, phenolic compounds in olive oil prevent lipoperoxidation, improve the lipid profile and endothelial function (inducing nitric oxide-mediated endothelium-dependent relaxation [29, 30]), and have antithrombotic properties [31].
The Food and Drug Administration (FDA) confirms that consuming approximately two tablespoons (23 g) of olive oil per day may reduce the risk of heart disease [5].
Another very famous part of the Mediterranean diet is linked to the consumption of red wine. Although alcohol consumption can cause multiple organ damage and raise BP, red wine consumption is inversely associated with mortality from cardiovascular diseases [32], risk reduction being greatest in the case of low to moderate intake [33, 34]. Studies performed with de-alcoholized red, but not white wine confirm short-term cardiovascular benefits [35]. This is because grape skins, seeds, and stems from which the red wine is produced [36] contain many bioactive polyphenols, including flavonoids (quercetin, catechin, and epicatechin), pro-anthocyanidins, and anthocyanins, phenolic acids (gallic, caftaric, and caffeic acid), and the trihydroxystilbene termed resveratrol.
More than a purely anti-hypertensive effect [37, 38], the red wine polyphenols improve endothelial function, leading to decreased arterial damage, decreased angiotensin-II activity, increased nitric oxide, decreased platelet aggregation, decreased LDL oxidation, and the stimulation of the anti-senescence factor sirtuin [35].
The Mediterranean diet is rich in fibers that are extremely effective in reducing both systolic and diastolic BP in hypertensive patients [39].
15.3.2.2 DASH Diet
The Dietary Approaches to Stop Hypertension (DASH) diet (Table 15.3) is an analog of the Mediterranean diet coded in the context of the US alimentation system, with the specific aim of lowering blood pressure.
Table 15.3
The Dietary Approaches to Stop Hypertension (DASH) diet
Food group | Daily servings | Serving sizes | Examples and notes | Significance of each food group to the DASH eating plan |
|---|---|---|---|---|
Grains and grain products | 7–8 | 1 slice bread 1 oz dry cereala ½ C cooked rice, pasta, or cereal | Whole wheat bread, English muffin, pita bread, bagel, cereals, grits, oatmeal, crackers, unsalted pretzels, and popcorn | Major sources of energy and fiber |
Vegetables | 4–5 | 1 C raw leafy vegetable ½ C cooked vegetable 6 oz vegetable juice | Tomatoes, potatoes, carrots, green peas, squash, broccoli, turnip greens, collards, kale, spinach, artichokes, green beans, lima beans, sweet potatoes | Rich sources of potassium, magnesium, and fiber |
Fruits | 4–5 | 6 oz fruit juice 1 medium fruit ¼ C dried fruit ½ C fresh, frozen, or canned fruit | Apricots, bananas, dates, grapes, oranges, orange juice, grapefruit, grapefruit juice, mangoes, melons, peaches, pineapples, prunes, raisins, strawberries, tangerines | Important sources of potassium, magnesium, and fiber |
Low-fat or fat-free dairy foods | 2–3 | 8 oz milk 1 C yogurt 1.5 oz cheese | Fat-free (skim) or low-fat (1%) milk, fat-free or low-fat buttermilk, fat-free or low-fat regular or frozen yogurt, low-fat and fat-free cheese | Major sources of calcium and protein |
Meats, poultry, and fish | 2 or less | 3 oz cooked meats, poultry, or fish | Select only lean; trim away visible fat; broil, roast, or boil instead of frying; remove skin from poultry | Rich sources of protein and magnesium |
Nuts, seeds, and dry beans | 4–5 per week | 1.5 oz or  C nuts C nuts½ oz or 2 Tbsp seeds ½ C cooked dry beans and peas | Almonds, filberts, mixed nuts, peanuts, walnuts, sunflower seeds, kidney beans, lentils | Rich sources of energy, magnesium, potassium, protein, and fiber |
Fats and oilsb | 2–3 | 1 tsp soft margarine 1 Tbsp low-fat mayonnaise 2 Tbsp light salad dressing 1 tsp vegetable oil | Soft margarine, low-fat mayonnaise, light salad dressing, vegetable oil (such as olive, corn, canola, or safflower) | In DASH 27% of the calories are fat, including that in or added to foods |
Sweets | 5 per week | 1 Tbsp sugar I Tbsp jelly or jam ½ oz jelly beans 8 oz lemonade | Maple syrup, sugar, jelly, jam, fruit-flavored gelatin, jelly beans, hard candy, fruit punch, sorbet, ices | Sweets should be low in fat |
As described by Plotnikoff and Dusek (cited in Rakel [5]): “the DASH diet trial enrolled 459 participants and provided each with all his or her food for 11 weeks. For the first 3 weeks, the participants were provided with a control diet that was low in fruits, vegetables, and dairy products, with a fat content typical of the average diet in the USA. Participants were then randomly assigned to receive the control diet, a diet rich in fruits and vegetables, or a “combination” diet rich in fruits, vegetables, and low-fat dairy products and with reduced saturated and total fat for 8 weeks. Sodium intake and body weight were maintained at constant levels. For the 326 participants with prehypertension, the DASH diet resulted in reduced SBP and DBP of 3.5 mmHg (P < 0.001) and 2.1 mmHg (P = 0.003), respectively. Among the 133 subjects with stage 1 hypertension, the DASH diet reduced SBP and DBP by 11.4 and 5.5 mmHg respectively, more than the control diet (P < 0.001 for each)” [40].
15.3.2.3 Anti-ACE Peptides
Observational and epidemiological studies demonstrate a consistent association between high protein intake and a reduction in BP [41]. The source of protein is important, being animal protein (milk, chicken, and fish, such as sardine [42] or bonito fish, a member of the tuna family [43]) less effective than non-animal protein [44] (such as soy, which contains isoflavones, saponins, phytic acid, amino acids, trypsin inhibitors, fiber, and globulins with an anti-hypertensive effect [45]).
15.3.2.4 Omega-3 Fatty Acids
A diet rich in omega-3 polyunsaturated fatty acids (contained in cold water fatty fish or grass-fed animals) may prevent the development of hypertension [46] and is an effective remedy in the early stages of the disease [47]. Omega-3 fatty acid deficiency contributed to the development of hypertension in animal models [48].
Furthermore, as the omega-3 fatty acids are effective at improving insulin sensitivity and dyslipidemia, they may be particularly useful in patients with the metabolic syndrome [1].
15.3.2.5 Cocoa (Theobroma cacao)
Regular intake of cocoa-containing foods is linked to lower cardiovascular and all-cause mortality and is inversely associated with BP [49, 50]. Regular cocoa (dark chocolate) intake demonstrated significant reductions in the values of both systolic and diastolic BP, in healthy subjects and in cases of prehypertension or grade 1 hypertension [5, 51].
In the studies cited, regular consumption of cocoa has not resulted in an increase in body weight, plasma levels of lipids, glucose, and 8-isoprostane (a measure of oxidative stress). The BP decrease was also accompanied by a sustained increase in the vasodilatory nitric oxide donor, S-nitrosoglutathione. In comparison, the polyphenol-free white chocolate intake caused no changes in BP or plasma biomarkers [51].
15.3.3 Supplements
The quality of the studies related to the possible beneficial effect of selective supplementation of certain elements is poor. This is because it is very difficult to determine whether the baseline value of an alleged deficient state is to trace in the blood, at an intracellular or simply at a functional level. Given the interindividual and intra-individual variability (for example, linked to the activation of the stress axis as a function of psycho-emotional states [3, 14, 15]), these forms of insufficiency can also be additive. Then, the question is that we often dose elements blindly: in some people, poor blood levels of a certain element can be traced back without this causing specific problems (the intracellular stores could be in order and functionally normal), whereas other people possess normal blood levels of the same element at the expense of the intracellular store or have functional deficits.
Therefore, it is difficult to establish a critical value of a nutrient that can be good for all people, below which supplementation is necessary. There is also individual variability in bioavailability, uptake, and metabolism that are often not considered in the available literature. The result is that much of the interventional nutrition research does not answer the very important clinical question, “Does replenishment of a deficiency to a given serum level result in an improved clinical outcome?” A summary of nutritional supplements in hypertension is presented in Table 15.4.
Supplements | Daily intake |
|---|---|
Coenzyme Q10 | 120–150 mg 2× a day |
Fish oil | 3–4 g a day |
Nattokinase (NSK-SD) | 50–100 mg if dietary natto is not consumed |
Magnesium | 400–800 mg a day |
Organic garlic | 1,000 mg if not taken in diet |
Hawthorn | 1,000 to 1,500 mg per day |
Quercetin | 500–1,000 mg per day 2× a day |
Vitamin D3 | 2000 units per day |
Vitamin B6 | 100 mg 1–2× a day |
Vitamin C | 250–500 mg 2× a day |
15.3.3.1 Potassium and Sodium
Diet low in potassium and high in sodium is linked to hypertension, cardiovascular diseases, and cancer [43]. Natural diets rich in fruit and vegetables provide the correct intake of potassium and sodium, although potassium supplementation with a good anti-hypertensive effect may be required [53–56]. Potassium should be used with caution in patients with kidney disease or those taking digitalis, potassium-sparing diuretics, and angiotensin-converting enzyme inhibitor anti-hypertensive drugs.
15.3.3.2 Magnesium
Magnesium homeostasis is so closely linked to that of potassium that some forms of hypokalemia result from a magnesium-deficient intake. Magnesium is a well-understood and frequently used intervention for the hypertension of preeclampsia. With a proper diet, such as the Mediterranean or DASH (natural sources of magnesium including pumpkin seeds, nuts, quinoa, spinach, bran cereal, buckwheat, and beans), the intake of magnesium should be sufficient not to require supplementation, as suggested by the JNC 7 guidelines [2].
However, several reports have demonstrated a link between higher intake of magnesium and lower blood pressure [57, 58]. On the other hand, low dietary intake of magnesium correlates strongly with high BP [59].
An important source of magnesium is water: water with many minerals such as magnesium is called “hard water” and there is an inverse correlation between water hardness and high blood pressure. Magnesium supplementation can be very beneficial in hypertensive subjects who take diuretics or who have high levels of renin. Magnesium may be administered in different formulations, but it seems to be better absorbed and more effective when linked to the intermediates of the Krebs cycle (aspartate, malate, succinate, fumarate, or citrate). It should keep in mind that serum magnesium levels do not reflect intracellular magnesium and the many forms of magnesium may have different bioavailability and physiological activity [60]. To quote Rakel: “The result is a ‘one-size-fits-all’ approach to magnesium studies of varying dosing and varying type of magnesium that prevents any meta-analysis of existing randomized trials. Despite these limitations, magnesium appears to be beneficial and nontoxic” [5].
15.3.4 Dosage
Starting at a low dose of the non-oxide forms of magnesium (120–200 mg/day) and slowly increase the dose to 400–1,200 mg/day) as tolerated, monitoring serum levels [54].
15.3.5 Precautions
Magnesium is well tolerated in general, but can sometimes cause looser stool; supplements must be used with great care in patients with kidney disease or severe heart disease (atrio-ventricular block) [1].
15.3.5.1 Calcium
Hypertension is linked to a low intake of calcium, but this evidence is not as strong as from magnesium or potassium [54, 61]. There are conflicting opinions [62] about the antihypertensive effect of calcium supplementation: it seems to be more effective in blacks, in salt-sensitive patients, and in elderly hypertensive patients [63, 64]. Calcium citrate is more effective than calcium carbonate.
15.3.5.2 Vitamins C, B6, and D
Vitamins C and B6 have a lowering BP effect in people with mild hypertension [43]. Vitamin C (with its antioxidant effect) promotes lead excretion, which is linked to hypertension and cardiovascular mortality [65–67]. Vitamin B6 supplementation (oral dosage 5 mg/kg) is linked to decreased serum norepinephrine [68, 69].
Calcitriol, also known as 1,25-dihydroxy vitamin D, is the activated form of vitamin D, controlling hundreds of genes, and an almost ubiquitous effect in the cardiovascular [70, 71] and renal system [72].
Although vitamin D deficiency is associated with an increased risk for hypertension [73, 74], its supplementation did not appear sufficient to be able to recommend its extensive use for the treatment of hypertension [75–77]. For this hormone, the considerations made at the beginning of the paragraph are of paramount importance. BP is finely regulated in a complex integrated system made of cytokines, hormones, and neurotransmitters [3, 14, 15]. It is difficult to establish a cutoff value to apply to all individuals (regardless of their psychological, emotional, and environmental characteristics), on which to base an effective supplementary intervention. As suggested by Plotnikoff and Dusek (cited in Rakel [5]): “the most important question at this time is whether a threshold serum level is needed to reduce the risk of incident hypertension or to reduce already elevated BPs” [5]. Additionally, the length of time of vitamin D sufficiency required for prevention or reduction must be defined.
15.3.5.3 Coenzyme Q10
Coenzyme Q10 (CoQ10, ubiquinone, or ubiquinol) is synthesized within the body, and is a crucial cofactor for the production of adenosine triphosphate (ATP), exerting a potent anti-oxidant effect. The highest tissue concentration is found in the heart, and the highest cellular concentration is on the inner membrane of the mitochondrion. Thirty-nine percent of hypertensive patients show a deficiency in this element [43, 78], as it occurs during aging or in the case of hyperthyroidism, cardiovascular disease, total parenteral nutrition, aerobic training, and ultraviolet exposure [79].
The supplementation of CoQ10 has the potential to significantly reduce both systolic and diastolic hypertension after 4–12 weeks of therapy [80], with concomitant improvements in cardiovascular functional status (New York Heart Association (NYHA) functional class) and medication requirements [81]. After an average of 4.4 months, 37% of patients were able to discontinue one antihypertensive drug, 11% discontinued two drugs, and 4% discontinued three drugs. Only 3% required the addition of one antihypertensive drug, and none required the addition of more than one antihypertensive drug. Twenty-five percent of all patients were able to control their BP with only CoQ10 supplementation [81] and had significant side effects [82]. In many studies, patients were able to discontinue medications [82].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


