NYHA class
Description
I
Physical activity not limited by symptoms such as shortness of breath, fatigue, or palpitations
II
Physical exertion mildly limited, with symptoms of shortness of breath, fatigue, or palpitations developing with typical daily activities
III
Physical activity severely curtailed; symptoms of shortness of breath, fatigue, or palpitations developing with any kind of activity
IV
Symptoms and physical discomfort present even at rest
The diagnosis is confirmed by echocardiography and cardiac catheterization.
Despite pharmacological and technological progress, morbidity, mortality, and the escalating financial burden to society associated with heart failure remain unacceptably high. At 40 years of age, the lifetime risk of developing heart failure for both men and women is 20%. A conservative estimate of the direct and indirect costs of heart failure in the USA for 2010 is $39.2 billion [7]. The 1-year mortality rate for heart failure is 20% [8, 9]. The incidence of heart failure is expected to increase with the increase in the average age of the population and survival following heart attacks [1].
With regard to the integrative medicine approach, the single best way to treat heart failure is to prevent its development and its inexorable vicious cycle, leading toward progressive greater infirmity and death within a few years. As stated by Russell and Greenfield (cited in Rakel [7]): “Prevention, prevention, prevention must be our mantra with respect to heart failure management. Do everything to prevent the disease from ever developing in the first place. Integrative means to help prevent or at least aggressively treat disorders that contribute to development of heart failure (including hypertension, coronary artery disease, diabetes, and dyslipidemia).”
It is essential to strengthen the educational interventions aimed at cardiovascular health early in life and carefully oversee and control the main cardiovascular risks, such as hypertension, diabetes, obesity, coronary artery disease, and stress [10–12].
As we will see, the integrated approach has great advantages in New York Heart Association (NYHA) classes I and II and in the early stages of heart failure (stages A to C in Table 17.2). For people who have already developed symptomatic heart failure, and in NYHA stages III and IV, the emphasis rests squarely on conventional medical therapy. Complementary medical therapies with the promise of efficacy and evidence for safety can be used as adjuncts, to the benefit of most patients.
Table 17.2
Stages of heart failure (HF)
ACC/AHA stage | Description |
|---|---|
A | At risk for HF, but without structural heart disease or HF symptoms |
B | Structural heart disease, but without signs or symptoms of HF |
C | Structural heart disease with prior or current symptoms of HF |
D | Refractory heart failure requiring specialized intervention |
Although the main objective of integrative cardiology is the prevention of heart failure, in patients with established heart failure progressive cardiovascular deterioration has to be prevented, symptoms minimized, quality of life enhanced, and survival rates increased.
In Fig. 17.1a and b the treatment algorithm for heart failure by stage of the disease is shown.
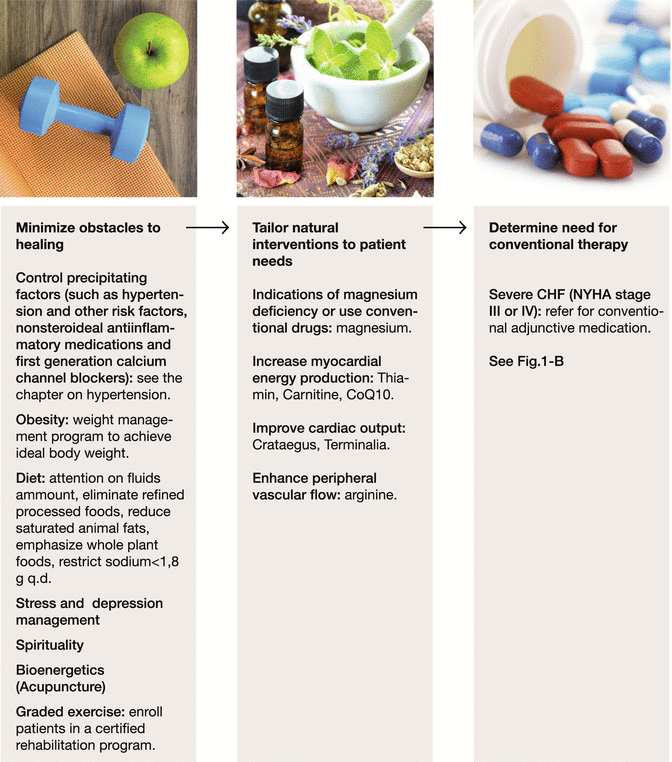
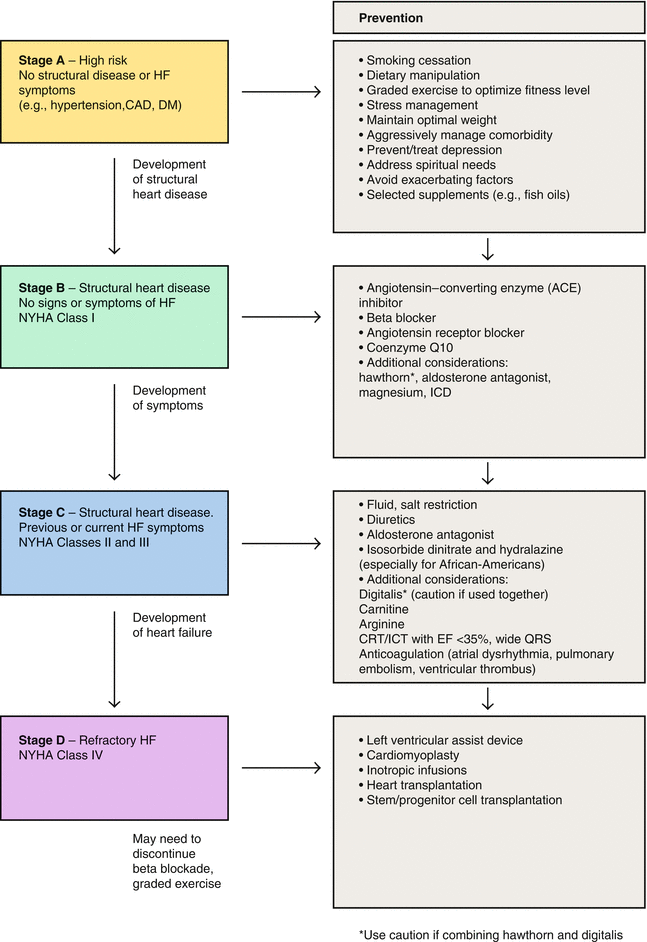


Fig. 17.1
(a) In summary: staged integrative approach to heart failure. Don’t smoke, follow an anti-inflammatory or Mediterranean diet, control weight, perform regular physical fitness activities, manage stress, treat depression, take care of spirituality, work to manage medical conditions that may lead to heart failure (high blood pressure, coronary artery disease, dyslipidemia, and diabetes), avoid overuse of nonsteroidal anti-inflammatory medications. (b) Clinical pathway: management of congestive heart failure (CHF). CAD coronary artery disease, CRT cardiac resynchronization therapy, DM diabetes mellitus, EF ejection fraction, HF heart failure, ICD implantable cardioverter defibrillator, NYHA New York Heart Association. Modified from Rakel [7]
17.2 Lifestyle and Nutrition
In addition to all the programs for prevention and care, especially in the context of CHF, close attention must be paid to tobacco and alcohol cessation and weight management, educating patients and their family regarding the adverse effects of smoking, excessive alcohol intake, and obesity. The heart is a muscle and needs to be trained: the condition of heart failure is not a contraindication to exercise. There is great deal of evidence [13–16] regarding the safety [17] and benefits of cardiological rehabilitation with graded exercise programming in patients with cardiac dysfunction in terms of functional improvement [18] and quality of life [19, 20]. The combination of exercise and proper diet helps to maintain a proper body weight by reducing the workload of a dysfunctional heart. Rest is important and patients should be advised to sleep at least 7–8 h per night [7, 21].
The doctors and professionals who care for patients with CHF should also pay attention to their spiritual side [22–25]. Indeed, many patients with CHF struggle with their spirituality, “a struggle that adds to an already stressful situation and perhaps leads to morbidity” [7, 26, 27]. The spiritual challenges associated with heart failure have been equated with those experienced by people with cancer [28]. As suggested by Russel and Greenfield (cited in Rakel [7]): “attention to spiritual needs can help people adjust to their new circumstances, address specific regrets with regard to prior lifestyle choices, and search for present meaning and future hope” [29, 30].
Dietary recommendations for hypertension are appropriate for most CHF patients. In particular, a Mediterranean or Dietary Approaches to Stop Hypertension (DASH) diet, low in sodium and high in potassium, paying attention to the ingestion of fluids, may help prevent development of heart failure and slow progression of established disease. Patients with CHF may need to limit sodium intake to 2 g/day and daily ingestion of water to 1.5–2 L.
17.3 Supplements
Nutritional supplements improve myocardial energy production and this is a very important effect in a pathological scenario always characterized by energy depletion, often due to nutrient or coenzyme deficiency [21].
17.3.1 Magnesium
Magnesium is a key element for adenosine triphosphate (ATP) production. In the case of CHF, magnesium levels correlate directly with survival rates [31] and deficiency is linked to arrhythmias, reduced cardiovascular prognosis, worsened ischemia, and increased mortality in acute myocardial infarction [1]. The deficiency of this mineral is probably caused by inadequate intake, increased wasting owing to the overactivation of renin–angiotensin–aldosterone system typical of CHF [32], and by conventional drug therapy such as digitalis, diuretics, beta-blockers, and calcium-channel blockers [33–36]. Magnesium supplementation produces positive effects in patients with CHF on conventional drugs, even if serum magnesium is normal [32]. The recommended dosage is: 200–400 mg t.i.d. (magnesium citrate).
17.3.2 Thiamine
Thiamine, or vitamin B1 deficiency, has remarkable cardiovascular effects, leading to the so-called wet beri-beri disease, a condition related to sodium retention, peripheral vasodilation, and heart failure [1]. A common important drug used in CHF treatment, such as furosemide, causes B1 deficiency [37], although it is uncommon to have a significant lack of thiamine, except in alcoholics [38].
Many people, especially elderly and hospitalized patients, however, do not reach the recommended daily dose of 1.5 mg [38]. A daily dose of 80–240 mg of vitamin B1 improves the clinical condition of CHF, as it improves patients’ ejection fraction by as much as 13–22% [39, 40]. Its use at doses of 200–250 mg qd, in the context of CHF, is recommendable, given the lack of risk and the low cost [21].
17.3.3 l-Carnitine
Carnitine is an important factor for the production of ATP, being a carrier of fatty acids into the myocardial mitochondria [21]. The myocardium stores considerable stocks of carnitine and coenzyme Q10, in excess of the metabolic standard needs, but this quantity is dramatically depleted in the course of ischemia and CHF [41–48]. Carnitine supplementation improves myocardial performance [47, 49–54] and outcome [55] in the case of CHF [56–59] in proportion to the duration of its use [60]: the longer it is used, the greater the improvement. When administered 500 mg t.i.d. for 6 months, the maximum exercise time improves by 16–25% and the ejection fraction by 12–13% [60]. When carnitine is given in an acute setting, it lowers pulmonary artery and capillary wedge pressures [59].
17.3.4 Coenzyme Q10
Coenzyme Q10 (CoQ10) is present in small amounts in most diets and is also synthesized within the body from tyrosine, partially through a common pathway shared with cholesterol synthesis. It is found in its highest concentrations within the mitochondrial membranes of organs that have significant energy requirements, especially the heart, where is involved with energy production [7, 61, 62]. CoQ10 exerts anti-oxidant and membrane-stabilizing effects as well [7, 63].
The amount of CoQ10 is reduced in patients with CHF and the deficit is greater, the worse the NYHA class [7, 64].
For CoQ10 the considerations made in the case of hypertension are also valid. In particular, it appears to be a key factor to be added to conventional CHF drugs [21, 65], because it improves the clinical picture and quality of life of patients with CHF [66–69]. In particular, the clinical signs of CHF (cyanosis, peripheral and pulmonary edema, hepatomegaly, venous congestion, palpitations, sweating, insomnia, dizziness, nocturia) improved by 50–80% with few and mild side effects [70]. Two recent studies failed to show clinical efficacy [71, 72].
The optimal dosage of CoQ10 in the setting of heart failure is as yet undetermined. Studies have used doses ranging from 30 to 600 mg/day, but most practitioners initially prescribe 100–200 mg daily. Softgel capsules of CoQ10 appear to provide superior bioavailability [73] and rare side effects (gastrointestinal upset) [74]. CoQ10 is reduced in those who take statins [75–79]. Care should be taken in the case of concomitant warfarin consumption [7, 80, 81], possibly necessitating dose adjustment.
17.3.5 l-Arginine
Arginine is a valuable amino acid in the treatment of CHF, improving hemodynamics and endothelial function [82–86], exercise tolerance [82, 87, 88], kidney function [89], and quality of life [90]. In fact, from arginine the body synthesizes nitric oxide with a vasodilatory effect. The administration of 5.6–12.6 g of arginine per day (mean dosage recommended 2–6 g [7]) leads to an increase in peripheral blood flow of 30% and an 8% increase in the distance traveled during the 6-min walking test [82].
17.3.6 Vitamin E
Questions persist about the safety of high-dose vitamin E in patients with established cardiovascular disease [91].
17.4 Phytotherapy
Herbs and supplements require in 4–6 weeks to obtain an evident clinical benefit and are useful in the early stages of heart failure (ACC/AHA stages A to C and NYHA classes I to III). Thus, the use of these agents is not appropriate for acutely worsening heart failure.
17.4.1 Hawthorn (Crataegus oxyacantha, laevigata, and monogyna)
The herbal medicinal plants and their derivatives are used as a complementary therapy for heart failure [92]; among these, in addition to those with diuretic and hypotensive activity, hawthorn plays an important role. The tops (flowers and leaves) of this plant are used and are obtained from the species Crataegus oxyacantha, laevigata, and monogyna.
Hawthorn (Crataegus oxyacantha) was considered a “cardio-sedative” folk medicine, but evidence proves that it has the opposite effect enough to be recommended in the complementary therapy of heart failure NYHA classes I and II (Table 17.3).
Increased coronary artery blood flow |
Enhanced pumping efficiency of the heart (improved contractility) |
Anti-oxidant activity |
Phosphodiesterase inhibition |
Angiotensin-converting enzyme inhibition |
Anti-dysrhythmic effects (lengthens the effective refractory period, unlike many cardiac drugs) |
Mild reduction in systemic vascular resistance (lowered blood pressure) |
Several pharmacological studies on hawthorn extract reveal a protective function on ischemic heart and myocardial damage related to hypertension. In particular, its flavonoids have an ACE inhibitory action with hypotensive effects [94, 96, 97] and protect the cardiomyocytes following ischemia-reperfusion [98]. Luteolin, hyperoside, vitexin, and rutin increase the coronary flow significantly, have a mildly positive inotropic effect, and optimize the speed of relaxation of heart myocells [99]. Anthocyanosides determine the liberation of PGI2, a prostaglandin with endothelial vasodilatory activity [100]. We recommend a dosage of 5–20 mg/day of flavonoids present in 250–1,000 mg/day of dry extract 2%.
In addition to its positive inotropic and lusitropic effects, the polyphenols, amines, and triterpenic acid content in hawthorn have a negative chronotropic action (reducing the heart rate), a positive dromotropic effect (reducing the conduction time), and a negative bathmotropic action (lengthening the refractory period) [101]. In arterial and coronary areas a decrease in peripheral and coronary resistance was observed, with a modest hypotensive action, an anti-oxidant, anti-inflammatory, and endothelial protective effect, and antiplatelet activity [100].
Different clinical trials featuring the hawthorn have followed over time [102, 103], confirming the effectiveness of the extracts of this plant in increasing the force of contraction of the heart, reducing the symptoms and dyspnea, and improving exercise tolerance, particularly in patients in NYHA classes I–II [28, 104–111]. In the various studies mentioned, two types of hawthorn extracts were employed, standardized in flavonoids to 2.2% or procyanidins 18.8%, with variable dosage from 160 mg to 1,800 mg/day. Most practitioners believe that therapeutic efficacy is greater with higher doses (600–1,800 mg/day) [7]. Again, no noticeable improvement may occur for 6–12 weeks.
Most of the studies with positive results, however, did not include treatment with drugs now accepted as standard medical therapy, such as ACE inhibitors and beta-blockers. Later studies employing hawthorn in the setting of CHF in combination with current standard medical therapy reported less successful outcomes [103, 112, 113]. Even in these studies a modest improvement in the left ventricular ejection fraction was identified along with a trend toward reduced cardiac mortality, most notably for those with significantly impaired left ventricular function [103, 112, 113].
Hawthorn is well tolerated and has few side effects (3% incidence) such as headache, fatigue, dizziness, palpitations, rash, drowsiness, agitation, and gastrointestinal disorders. It may interact with drugs such as digitalis, beta-blockers, diuretics, ACE inhibitors, and platelet anti-aggregants [114]. Nevertheless, the results of a retrospective safety analysis should be reported, where hawthorn use not only failed to impede the progression of the disease, but also appeared in some patients to increase the risk of early heart failure progression. In light of a previously good safety record, these findings are puzzling and concerning. Probably, the use of hawthorn in a holistic program that includes nutritional care and adequate stress management could avoid these outcomes [10–12].
In future, as suggested by Russel and Greenfield (cited in Rakel [7]): “because the purported beneficial actions of hawthorn overlap some of those inherent to medications such as ACE inhibitors and beta blockers, combination therapy possibly would permit the use of lower doses with no diminution of therapeutic effectiveness.”
Nowadays, the digital and pharmaceutical formulations based on hawthorn find elective indications in patients with impaired myocardial contractile force only in the case of atrial fibrillation; otherwise, the mortality of patients with heart failure increases [1]. Most conventional medical practitioners reflexively state that hawthorn should not be given to people taking digitalis for heart failure [115]. In the past, sparteine, an extract of the broom (Sarothamnus scoparius), was also used, but then abandoned for toxicity reasons [100].
17.4.2 Terminalia arjuna
Terminalia arjuna is a traditional Ayurvedic herb that has recently been described to be effective for heart failure in a controlled clinical study [116]. The dosage is 500 mg every 8 h of the extract from its bark. The study documents a very promising effect of the plant in the case of severe and refractory heart failure with improvements to NYHA class II.
17.4.3 Tricks of the Trade
Undoubtedly, the plant most used in the case of heart failure is the hawthorn with excellent results when associated with ginseng, which enhances its tonic properties. To avoid hawthorn’s “opposite effect” it is better to use it in the form of glycerine macerate (GM): using the gems, in fact, the action seems to be more “regulatory” compared with its leaves and flowers. For example: hawthorn tincture lowers blood pressure, whereas hawthorn GM rules it (that is, in the case of low blood pressure, it tends to raise the blood pressure a little). Hawthorn tincture works on tachycardia, whereas hawthorn GM also works on bradycardia. In addition, hawthorn MG can safely be combined with drugs.
Another great remedy that is used in the naturopathic field for both the prevention and treatment of infarction, heart failure, and for rhythm disturbances (angina, arrhythmias, extrasystoles, extrasystoles with mitral valve prolapse) is the dietary supplement magnesium–potassium–bromelain: 1 tablet three times a day. Bromelain is an enzyme derived from the pineapple stem and has a significant anti-inflammatory action on vascular walls combined with a lipid-lowering effect. To obtain good results, it should be taken for at least 6 months.
17.5 Bioenergetics (Acupuncture)
Acupuncture, modulating sympathetic activity, may be useful for people with heart failure [117, 118]. A pilot study of acupuncture reported intriguing results documenting a marked improvement in the 6-min walk test in subjects with stable NYHA class II to III heart failure on appropriate medical therapy compared with controls [119]. More research is needed to establish the precise rules for the application of this method in the context of CHF.
17.6 Mind–Body Therapies
In the clinical CHF context, the mind–body therapies play an important role in growing success. In particular, they seem to be very useful in combating depression that often accompanies the life of patients with heart failure [120, 121]. Depression is an independent negative prognostic factor in CHF and even alone can determine inflammation [122, 123], oxidative stress, and endothelial dysfunction with possible myocardial ischemia, leading to increased morbidity and mortality [10–12]. Numerous studies describe that an appropriate reduction in stress, in patients with CHF, causes a relief of depression and anxiety [124] with an improvement in risk profile and cardiovascular disease progression and outcomes [125–131]. Specifically, are seen an improvement of the autonomic balance and of the levels of circulating neurotransmitters [132], an increase in cardiac output and a decrease in peripheral resistance [133], and an increase in exercise capacity, a decrease in hospitalizations, with improvement of symptoms [134]. The techniques that can be applied in a program of care for a patient with CHF are: biofeedback [133], mindfulness meditation [135], and transcendental meditation [134]; other studies regarding the beneficial effect of mind–body therapies involve freeze-frame stress management [136], relaxation response training [137, 138], tai chi [139, 140], and behavioral changes [141].
17.7 Pharmaceuticals, Biomechanical therapy, Surgery
According to international cardiological guidelines, all patients with heart failure should be started on some combination of angiotensin-converting enzyme inhibitor, angiotensin receptor blocker, beta-blocker, and aldosterone antagonists, which all have a positive impact on mortality related to heart failure [1, 12]. Diuretics act by decreasing the preload, lessening the cardiac workload, and the most commonly used are loop diuretics, such as furosemide, which are especially beneficial once congestion has developed. Isosorbide dinitrate and hydralazine are useful for the treatment of heart failure in African–Americans (who do not appear to respond as favorably to ACE inhibitors or beta-blockers).
The acute setting may require the infusion of inotropes, ultrafiltration, or aortic counterpulsation.
The most significant recent change in the conventional medical treatment of CHF is the increased reliance on device therapy (cardiac resynchronization therapy and implantable cardioverter defibrillators) and left ventricular assist devices. For indications for their use, see Camm et al., McMurray et al., and Tarzia et al. [1, 142–144]. Heart transplantation remains the gold standard in the case of refractory heart failure.
17.8 Future Therapies
Future therapies under study for heart failure [145, 146] treatment involve: calcium sensitizers, continuous-flow left ventricular assist devices, cytokine inhibitors, endothelin receptor blockers, erythropoiesis-stimulating proteins, fish oils, free fatty acid oxidation inhibitors, gene expression (miRNA), matrix metalloproteinase inhibitors, modified natriuretic peptides, nitric oxide–enhancing therapy, phosphodiesterase-III inhibitors, ribose, statin therapy, stem and progenitor cell transplantation, taurine, and vasopressin antagonists.
References
1.
Camm AJ, Thomas F, Luscher PWS (2009) ESC textbook of cardiovascular medicine, 2nd edn. Oxford Med: 1–1398
2.
Uddin N, Patterson JH (2007) Current guidelines for treatment of heart failure: 2006 update. Pharmacotherapy 27:12S–17SPubMed
3.
Young JB (1995) Contemporary management of patients with heart failure. Med Clin North Am 79:1171–1190PubMed
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


