This study aimed to analyze the impact of body mass index (BMI) and the metabolic syndrome (MS) on responses to clopidogrel or prasugrel and bleeding risk after acute coronary syndrome. The study included 1,542 consecutive patients who underwent coronary stenting (287 clopidogrel 75 mg, 868 clopidogrel 150 mg, and 387 prasugrel 10 mg). Platelet reactivity was assessed 1 month after discharge using platelet reactivity index vasodilator stimulated phosphoprotein (PRI VASP). Three hundred thirty-six patients (21.8%) were obese (BMI ≥30), and we observed higher platelet reactivity associated with higher BMI across thienopyridine regimens. Incidence of high on-treatment platelet reactivity (PRI VASP >50%) was higher in obese than nonobese patients (p <0.05 for all regimens). Conversely, incidence of low on-treatment platelet reactivity with prasugrel therapy (PRI VASP <20%) was lower in obese than nonobese patients: 13% (12 of 93) versus 33% (97 of 294); odds ratio 0.30, 95% confidence interval 0.16 to 0.58; p <0.001. Accordingly, incidence of Bleeding Academic Research Consortium bleeding complications was higher in nonobese than in obese patients: 10% (119 of 1,206) versus 6% (20 of 336); odds ratio 1.7, 95% confidence interval 1.1 to 2.8; p = 0.03. This impaired response was only observed in obese patients with the MS, and obese with the MS had significantly higher platelet reactivity than other obese patients with all regimens (p <0.01). Obese patients without the MS had no significant difference in platelet reactivity compared with nonobese patients. In conclusion, the present study confirmed that BMI has a strong impact on response to clopidogrel and prasugrel with higher incidence of high on-treatment platelet reactivity, lower incidence of low on-treatment platelet reactivity, and lower bleeding complication in obese patients. However, among obese patients, the presence of the MS strongly affects response to antiplatelet agents, indicating that the metabolic status might be a better predictor of platelet inhibition than BMI.
Dual antiplatelet therapy combining aspirin and clopidogrel has been the gold standard after acute coronary syndrome. However, despite the use of this strategy, recurrent events occur, and approximately 10% of patients will present an ischemic event after acute coronary syndrome. Variability of response to clopidogrel has been proposed as a responsible factor for these unwanted events. The study of platelet function under clopidogrel showed a linear correlation between platelet inhibition level and occurrence of complications for both ischemic and bleeding events. More recently, several studies have shown a significant association between bleeding and platelet reactivity in patients treated with prasugrel. A few studies have suggested obesity as an independent factor that intervenes negatively in clopidogrel responsiveness. These studies, however, assessed exclusively clopidogrel and only the effect of a loading dose (300 to 600 mg) rather than of longer-term treatment. Also, low body weight has been identified as a predictor of better response to prasugrel and a higher rate of bleeding complications in the trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel (TRITON) study. Obese patients exhibit an unfavorable cardiovascular prognosis compared with the general population. The efficacy and tolerance of the latest generation of thienopyridines have not been studied in this population specifically. Our study therefore aimed to evaluate, using platelet reactivity index (PRI) vasodilator stimulated phosphoprotein (VASP), the biological efficacy of the different antiplatelet treatments (clopidogrel 75 or 150 mg and prasugrel 10 mg) on a long-term basis in obese patients (body mass index [BMI] >30), as well as any potential association with ischemic or hemorrhagic events, compared with nonobese patients. This study was also intended to assess the impact of the metabolic syndrome (MS) in obese patients regarding response to antiplatelet agent.
Methods
Between July 2008 and December 2012, all patients who were admitted to our center for stable coronary artery disease or acute coronary syndrome (unstable angina, ST-segment elevation myocardial infarction, or non-ST-segment elevation myocardial infarction) and who underwent coronary angioplasty were consecutively enrolled. The patients were discharged with a maintenance dose of 75 mg of clopidogrel, 150 mg of clopidogrel, or 10 mg of prasugrel in combination with 75 mg of aspirin. The choice of the dual-therapy class was left to the physician’s discretion. Patient’s BMI was calculated using the standard weight/height 2 formula. The patients were divided into 2 groups depending on BMI: obese (BMI ≥30) or nonobese (BMI <30). The obese population was further divided in groups according to the international obesity classification: class 1 obesity (30 kg/m 2 ≤ BMI <34.9), class 2 (35 ≤ BMI <39.9), and class 3 (BMI ≥40). Patient inclusion criteria were admission for acute coronary syndrome or stable coronary artery disease; having undergone PCI; and receiving dual antiplatelet therapy on hospital discharge, which combined aspirin 75 mg with a maintenance dose of clopidogrel 75 mg, clopidogrel 150 mg, or prasugrel 10 mg. Patient exclusion criteria were antecedents of gastrointestinal or cerebral bleeding, creatinine clearance <25 ml/min according to the Cockcroft formula, or thrombocytopenia <100 G/L, which is a contraindication to antiplatelet therapy. Adherence was systematically assessed. The ethics committee of our institution approved the study protocol, and patients gave written informed consent for participation. Blood samples were drawn at 1-month follow-up, and VASP phosphorylation state of whole blood was assessed by flow cytometric assay [Platelet VASP, Diagnostica Stago (Biocytex), Asnières, France]. PRI VASP was calculated according to the formula: PRI VASP = [(MFI (PGE1) − MFI (PGE1 + ADP) /MFI PGE1 ] × 100. High on-treatment platelet reactivity (HTPR) was defined as PRI VASP >50% as previously proposed and low on-treatment platelet reactivity (LTPR) as PRI VASP <20% as described by our group. Obese patients were classified into 2 groups in which platelet reactivity were compared: obese without metabolic disorder according to Third Report of the National Cholesterol Education Program and obese with MS determined by ≥3 of the following 5 criteria: waist circumference (>102 cm for men and >88 cm for women), hypertriglyceridemia (triglycerides ≥1.7 mmol/L), low high-density lipoprotein cholesterol (<1 mmol/L for men and <1.3 mmol/L for women), fasting glucose ≥6.1 mmol/L, blood pressure ≥130 and/or ≥85 mm Hg. The biochemical parameters were measured in blood samples using different autoanalyzers; Beckman DX 800 (Beckman Coulter, Inc., Fullerton, California) for serum lipids, glucose, and creatinine. Clinical follow-up was planned for all patients at 1 month after hospital discharge with clinical visit and platelet function testing was at 1 month only. The clinical end points was the occurrence of bleeding events according to the Academic Bleeding Consensus Definitions with type 1, 2, or 3 or 5 (Type 4 was not expected because no patient had planned coronary artery bypass grafting). Although radial access is widely used in our center and access-site bleedings are less dependent on platelet inhibition, we collected only the non-access-site bleeding complications. Hard ischemic end points were also collected, and the primary ischemic end point was occurrence of definite or probable stent thrombosis.
The primary end points of the present study were defined as
- •
response to thienopyridine using PRI VASP in obese versus nonobese patients and in different classes of obesity,
- •
incidence of HTPR (PRI VASP >50%) in obese versus nonobese patients and in different classes of obesity,
- •
incidence of LTPR (PRI VASP <20%) in obese versus nonobese patients and in different classes of obesity, and
- •
incidence of bleeding and ischemic events in obese versus nonobese patients.
Statistical analyses were performed using PASW Statistics version 17.0 (SPSS Inc., Hong Kong). Continuous variables were reported as means and SD or as medians and range (according to their distribution), and categorical variables were reported as count and percentages. Univariate and multivariate analyses were performed using a logistic regression model. One-way analysis of variance was used for comparison between different groups. Odds ratios (ORs) were estimated with a 95% confidence interval (CI). Calibration of the logistic model was assessed using the Hosmer-Lemeshow goodness-of-fit test. For all tests, statistical significance was defined as p <0.05.
Results
During the study period, 1,542 patients were enrolled, of which 287 were treated with clopidogrel 75 mg, 868 with clopidogrel 150 mg, and 387 with prasugrel 10 mg. Among these patients, 24 (1.7%) were underweight (BMI <18.5), 521 (33.4%) normal weight, 661 (43%) overweight (26 ≤ BMI <30), and 336 (21.8%) were classified as obese (BMI ≥30). Of these, 264 (78.6%) were defined as class 1, 57 (17%) as class 2, and 15 (4.4%) as class 3. The clinical characteristics of the 2 patient groups are summarized in Table 1 . The obese patients were significantly younger than the nonobese patients (62.5 ± 10 vs 65 ± 12 years, p <0.01). Likewise, hypertension was significantly more common in the obese patients (65% vs 55%, p <0.01), as were diabetes (47.6 vs 25.3%, p <0.01) and dyslipidemia (65.5 vs 52.4%, p <0.01), compared with nonobese patients. Metabolic characteristics of the 2 groups are summarized in Table 2 . The obese patients displayed a higher degree of insulin resistance status than the nonobese patients did, as shown by significantly higher insulinemia, fasting glycemia, and glycated hemoglobin levels (p <0.01 for all). Similarly, triglyceride levels were significantly higher in obese patients (1.48 vs 1.18 g/L, p <0.01), as was lower high-density lipoprotein cholesterol (0.38 vs 0.42 g/L, p <0.01).
| Variable % or Mean ± SD | Nonobese Group (n = 1,206) | Obese Group (n = 336) | p (Mann-Whitney) |
|---|---|---|---|
| Age (yrs) | 65 ± 12 | 62.5 ± 10 | <0.01 |
| Female gender | 17.9% | 26.6% | 0.02 |
| Clinical presentation | |||
| Stable angina pectoris | 12.8% | 10% | NS |
| NSTEMI | 24.7% | 33% | <0.01 |
| Unstable angina pectoris | 30.9% | 35.1% | <0.01 |
| STEMI | 31.8% | 21.6% | <0.01 |
| CV risk factors | |||
| Previous acute coronary syndrome | 33.8% | 45.1% | 0.02 |
| Hypertension | 55% | 65% | <0.01 |
| Diabetes mellitus | 25.3% | 47.6% | <0.01 |
| Smoker | 38.5% | 39% | NS |
| Dyslipidemia | 52.4% | 65.5% | <0.01 |
| Family history | 26% | 27% | NS |
| Ejection fraction (%) | 55 ± 8 | 55 ± 7 | NS |
| Number of affected vessels | 1.7 | 1.8 | NS |
| Nonobese Group (n = 1,206) | Obese Group (n = 336) | p (Mann-Whitney) | |
|---|---|---|---|
| Fasting insulinemia (mIU/L) | 11.7 | 19.54 | <0.01 |
| Fasting glycemia (mmol/L) | 6.23 | 7.05 | <0.01 |
| C-reactive protein (mg/L) | 5.11 | 5.99 | <0.01 |
| Triglycerides (g/L) | 1.18 | 1.48 | <0.01 |
| Total cholesterol (g/L) | 1.5 | 1.49 | NS |
| HDL cholesterol (g/L) | 0.42 | 0.38 | <0.01 |
| LDL cholesterol (g/L) | 0.85 | 0.82 | NS |
| HbA1c (%) | 6.22 | 6.63 | <0.01 |
| Hemoglobin (g/dl) | 13.7 | 14.09 | NS |
| Platelets (g/L) | 250 | 248 | NS |
| Creatinine (mμmol/L) | 95.88 | 94.12 | 0.03 |
| Fibrinogen (g/L) | 4.45 | 3.97 | NS |
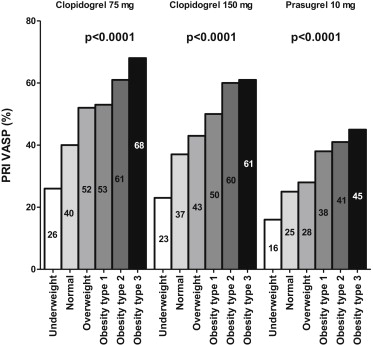
Obese patients were classified according to MS, and we identified 66% obese patients with metabolic syndrome (n = 222) and 34% without metabolic syndrome (n = 114). The impact of BMI on thienopyridine responsiveness was analyzed using the PRI VASP method; results are summarized in Figure 1 . On a clopidogrel 75 mg, clopidogrel 150 mg, or prasugrel 10 mg maintenance dose, the response to antiplatelet therapy was clearly and statistically correlated with BMI (p <0.01). The higher a patient’s BMI, the more residual platelet reactivity under treatment there was, regardless of long-term treatment regimen prescribed. Figure 2 summarizes the incidence of HTPR according to the 2 groups. Significantly more patients exhibited a PRI VASP >50% in the obese than in the nonobese group regardless of the treatment regimen prescribed (clopidogrel 75 mg: 63% vs 38%, p <0.01; clopidogrel 150 mg: 61% vs 31%, p = 0.04; prasugrel 10 mg: 28% vs 4%, p <0.01). The superior efficacy of prasugrel was emphasized by a significantly lower HTPR rate than that observed in clopidogrel patients (p <0.01). LTPR (PRI VASP <20%) on prasugrel therapy was significantly more frequent in nonobese than in obese patients (OR 0.30, 95% CI 0.16 to 0.58, p <0.001). Interestingly, among the obese patients, we observed a strong impact of metabolic syndrome on response to thienopyridine with all regimens with significantly impaired response and higher platelet reactivity in obese with metabolic syndrome compared with obese without MS ( Figure 3 ). Interestingly, obese patients without MS had no significant difference in platelet reactivity with all drugs compared with nonobese patients ( Figure 3 ). This difference remains significant after adjustment with confounding variables including BMI. For all drug regimens, in multivariate analysis, metabolic syndrome was a stronger predictor of HTPR than obesity itself. Nine percent of patients (139 of 1,542) suffered from bleeding complications during 6-month follow-up, including 67 Bleeding Academic Research Consortium (BARC) 1, 52 BARC 2, and 20 BARC 3 bleedings, with 8 life-threatening and 3 intracranial bleeding events. No significant difference was observed for occurrence of stent thrombosis between obese and nonobese patients, nor was there a significant association between platelet reactivity and stent thrombosis. Conversely, statistically more bleeding episodes (as defined by the BARC classification) occurred in nonobese patients than in obese patients (6% [20 of 336] vs 10% [119 of 1,206], OR 1.7, 95% CI 1.1 to 2.8, p = 0.03). This may be accounted for by the fact that there were more hyper-responders in the nonobese group but more hyporesponders in the obese group. Indeed, in the entire population, patients with bleeding had lower PRI VASP values than patients without bleeding: 27% ± 17% versus 41% ± 19%, p <0.001.