Chapter 78 Hepatic and Biliary Disease
The pulmonary consequences of liver disorders can be classified according to whether they predominantly affect the pleural space, the lung parenchyma, or the pulmonary circulation. Unique pulmonary abnormalities occur in patients with severe alpha1-antitrypsin deficiency, primary biliary cirrhosis (PBC), and primary sclerosing cholangitis (PSC). The impact of hepatic dysfunction on the respiratory system during sleep has recently been appreciated (Table 78-1).
Table 78-1 Major Pulmonary Consequences of Portal Hypertension (with or Without Cirrhosis)
| Area/Disorder | Complications |
|---|---|
| Pleural space | |
| Pulmonary parenchyma | |
| Pulmonary circulation | |
| Sleep-disordered breathing |
HCV, hepatitis C virus; NSIP, nonspecific interstitial pneumonia; PBC, primary biliary cirrhosis; PSC, primary sclerosing cholangitis; SaO2, arterial oxygen saturation; UIP, usual interstitial pneumonia.
Pathophysiology
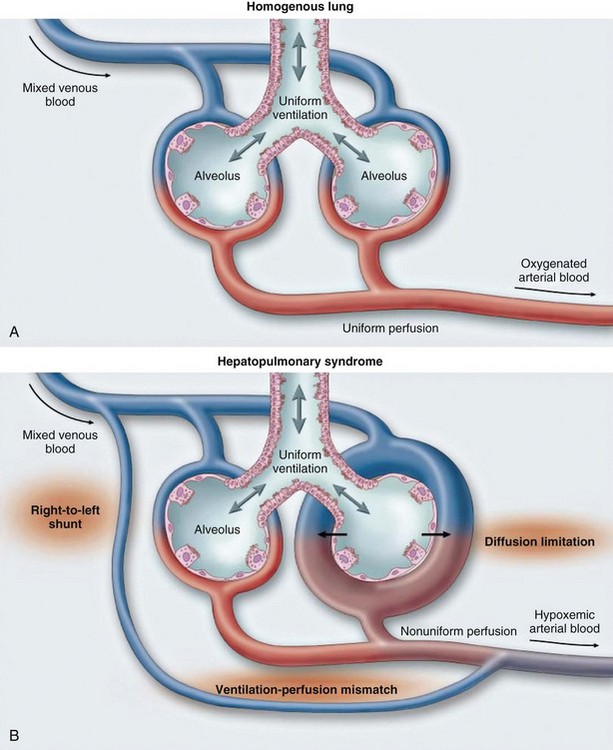
Hepatopulmonary Syndrome
HPS is characterized by arterial hypoxemia caused by precapillary-capillary dilations or direct arteriovenous communications. Excess perfusion for a given area of ventilation, diffusion limitation, and true anatomic shunting contribute to the degree of abnormal arterial oxygenation (Figure 78-1). Increased concentrations of exhaled nitric oxide have been demonstrated in HPS. Animal models of HPS have found upregulation of endothelin B receptors causing vasodilation and evidence of angiogenesis.
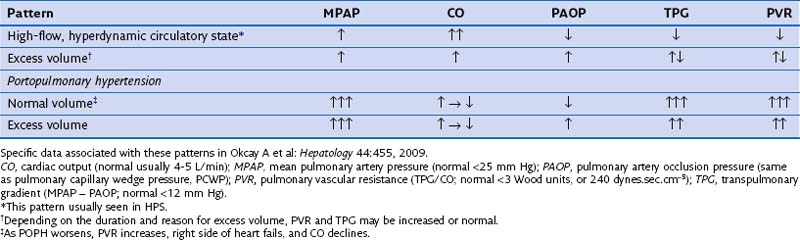
Portopulmonary Hypertension
POPH results from vasoproliferation (endothelium and smooth muscle), along with in situ thrombosis, which causes an increased pulmonary vascular resistance (PVR) to arterial flow. It is important to note the various pulmonary hemodynamic patterns that may exist in the patient with advanced liver disease (Table 78-2).