The CHADS 2 scoring system was found to be a good predictor for risk stratification of stroke in patients with atrial fibrillation. The effectiveness of this scoring system in assessing thrombogenic milieu before direct-current cardioversion has not yet fully been established on a large scale. In this study, data from 2,369 consecutive patients in whom transesophageal echocardiography was performed for screening before direct-current cardioversion from 1999 to 2008 were analyzed. Left atrial (LA) or LA appendage (LAA) thrombogenic milieu (spontaneous echo contrast, sludge, and thrombus) was investigated. The results were correlated with CHADS 2 score findings. The mean age was 66 ± 13 years, and the ratio of men to women was 2.2:1. CHADS 2 scores of 0, 1, 2, 3, 4, 5 and 6 were present in 11%, 25%, 30%, 22%, 8%, 3%, and 1% of the studies, respectively. The prevalence of LA or LAA sludge or thrombus increased with increasing CHADS 2 scores (2.3%, 7%, 8.5%, 9.9%, 12.3%, and 14.1% for scores of 0, 1, 2, 3, 4, and 5 or 6, respectively, p = 0.01). In a multivariate model, an ejection fraction ≤20% was the best predictor of LA or LAA sludge or thrombus (odds ratio 2.99, p <0.001). In conclusion, transesophageal echocardiographic markers of thrombogenic milieu were highly correlated with increasing CHADS 2 scores in patients who underwent transesophageal echocardiography–guided cardioversion. Giving more value to echocardiographic findings, such as the left ventricular ejection fraction, and its different levels (especially an ejection fraction ≤20%) might improve the precision of the CHADS 2 scoring scheme to predict thrombogenic milieu in the left atrium or LAA as a surrogate to cardioembolic risk in patients with atrial fibrillation.
The CHADS 2 scoring system has been widely used for general stroke risk stratification; however, to our knowledge, the capability of this scheme to differentiate cardioembolic versus noncardioembolic stroke risk has not been completely investigated. The aim of this study was to evaluate the effectiveness of the CHADS 2 scoring system to identify cardioembolic risk in patients with atrial fibrillation (AF) who undergo transesophageal echocardiography (TEE)–guided direct-current cardioversion (DCC). Additionally, we assessed the correlation strength of the different components of the CHADS 2 score to left atrial (LA) or LA appendage (LAA) features of thrombogenic milieu (spontaneous echo contrast [SEC], sludge, and thrombus) detected by TEE.
Methods
We identified 2,705 consecutive patients from January 1999 to December 2008 who underwent a TEE at the Cleveland Clinic for screening LA or LAA thrombogenic milieu (SEC, sludge, and thrombus) before DCC. Of those, 336 patients were excluded because of histories of rheumatic heart disease, prosthetic valve replacement, and significant (moderate or severe) valvular diseases. These data were extracted from the institution’s echocardiography and electrophysiology databases and then analyzed. The study was approved by the Cleveland Clinic Institutional Review Board.
A CHADS 2 score ranging from 0 to 6 was measured for each patient at the time of TEE. Scoring was done as follows: congestive heart failure = 1 point, hypertension = 1 point, age >75 = 1 point, diabetes mellitus = 1 point, and history of transient ischemic attack (TIA), stroke, or peripheral embolism = 2 points. A CHADS 2 score of 0 or 1 was defined as indicating low thromboembolic risk.
The history and duration of anticoagulation and antiplatelet therapy were reviewed. Paroxysmal AF was defined as recurrent AF (≥2 episodes) that terminated spontaneously within 7 days. Persistent AF was defined as AF that was sustained beyond 7 days or that lasted <7 days with the need to be cardioverted either pharmacologically or electrically.
TEE was performed using commercially available probes equipped with a multiplane phased-array transducer with a frequency range of 3 to 7 MHz within the 24 hours before DCC. Images of the left atrium and LAA were acquired at the midesophageal level, with rotation of the transducer array around multiple planes (0°, 45°, 90°, and 135°).
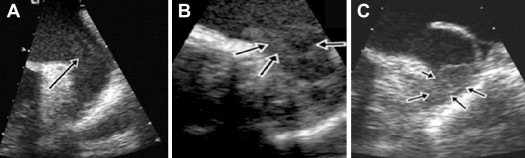
All transesophageal echocardiographic study reports were reviewed by observers blinded to the clinical histories of the patients. Transesophageal echocardiographic findings were categorized as follows: LA or LAA SEC was defined as “smokelike” echoes with the characteristic swirling motion with optimal gain setting during the cardiac cycle ( Figure 1 ). SEC was graded as mild or severe according to the following: near gain was increased progressively to just below the level at which static background noise appeared (threshold gain). Mild SEC was undetected at low gain and was seen in some parts of the left atrium or the entire left atrium at the threshold gain. Severe SEC occupied the entire atrium and appeared very dense even at low gain. Sludge was described as a dynamic gelatinous, precipitous echo density without a discrete mass that was present throughout the cardiac cycle ( Figure 1 ). Thrombus was defined as a circumscribed and uniformly echo-dense intracavitary mass distinct from the underlying LA or LAA endocardium and pectinate muscles which was present in >1 imaging plane ( Figure 1 ).
To assess inter- and intraobserver variability, a nested case group of 20 patients from the TEE database, including normal findings, SEC, sludge, and thrombus (5 each), was randomly selected and analyzed by the 2 same observers at 2 points 4 weeks apart. Intra- and interobserver concordance was very high in the identification of sludge and thrombus (κ ∼ 1.00) and was previously reported.
The transesophageal echocardiographic characteristics of thrombogenic milieu (SEC, sludge, and thrombus) and the mean left ventricular ejection fraction (LVEF) were compared across different levels of CHADS 2 scores using chi-square tests. In addition, a multivariate logistic regression model was used to identify independent clinical predictors of LA or LAA sludge or thrombus. The variables in the model were congestive heart failure, hypertension, an ejection fraction ≤20%, age >75 years, diabetes mellitus, and history of stroke, TIA, or systemic emboli. The model was examined with use of an ejection fraction ≤30% and ≤40% as well. In addition, the international normalized ratio and the duration of anticoagulation before cardioversion were assessed as confounders in the aforementioned model.
A p value <0.05 was considered statistically significant. Statistical analysis was done using Stata version 11 (StataCorp LP, College Station, Texas).
Results
Patient demographic characteristics are listed in Table 1 . We analyzed 2,369 patients (mean age 66 ± 13 years), 69% of whom were man (male-to-female ratio 2.2:1). CHADS 2 scores of 0, 1, 2, 3, 4, 5, and 6 were present in 11%, 25%, 30%, 22%, 8%, 3%, and 1% of the studies, respectively ( Table 1 ). Almost 1/3 of the cases (36%) belonged to the low-risk group (score 0 or 1). The prevalence of LA or LAA SEC was 49% (1,169 patients; 38% mild and 11% severe; Table 1 ). Sludge and thrombus were seen in 2% (n = 46) and 6% (n = 149) of patients, respectively.
| Characteristic | Value |
|---|---|
| Age (yrs) | 66 ± 13 |
| Men | 1,637 (69%) |
| Body mass index (kg/m 2 ) | 30.5 ± 14 |
| Heart failure | 1,415 (60%) |
| Coronary artery disease ∗ | 641 (27%) |
| Hypertension | 1,568 (66%) |
| Diabetes mellitus | 610 (26%) |
| Stroke/TIA/systemic embolic events | 236 (10%) |
| LVEF (%) | 42 ± 15 |
| Previous pulmonary vein isolation | 92 (4%) |
| Previous cardioversion | 242 (22%) |
| Previous pulmonary vein isolation and cardioversion | 80 (3%) |
| Anticoagulation at the time of cardioversion | |
| Warfarin | 806 (34%) |
| Heparin infusion | 1,421 (61%) |
| Low–molecular weight heparin | 75 (3%) |
| Bivalirudin infusion | 47 (2%) |
| Baseline anticoagulation before cardioversion | |
| Warfarin | 1,890 (80%) |
| Aspirin only | 232 (10%) |
| Clopidogrel only | 27 (1%) |
| None or not reported | 222 (9%) |
| Duration of anticoagulation before cardioversion (days) | |
| <2 | 529 (22%) |
| 3–14 | 607 (26%) |
| 15–28 | 50 (2%) |
| >28 | 641 (27%) |
| Not reported | 542 (23%) |
| CHADS 2 score | |
| 0 | 269 (11%) |
| 1 | 602 (25%) |
| 2 | 721 (30%) |
| 3 | 514 (22%) |
| 4 | 178 (8%) |
| 5 | 67 (3%) |
| 6 | 18 (1%) |
| LA/LAA SEC | 1,169 (49%) |
| Mild | 899 (38%) |
| Severe | 270 (11%) |
| LA/LAA sludge | 46 (2%) |
| LA/LAA thrombus | 149 (6%) |
| Type of baseline arrhythmia | |
| AF | 1,648 (70%) |
| Atrial flutter | 355 (15%) |
| Both AF/atrial flutter | 315 (13%) |
| Atrial tachycardia | 31 (1%) |
| Not reported | 21 (1%) |
∗ History of myocardial infarction, revascularization, or angiography with evidence of coronary artery disease.
The prevalence of LA or LAA thrombus or sludge increased with increasing CHADS 2 score (2.3%, 7%, 8.5%, 9.9%, 12.3%, and 14.1% for scores of 0, 1, 2, 3, 4, and 5 or 6, respectively, p = 0.01; Figure 2 ). SEC (total, mild, and severe) prevalence also increased significantly along with increasing CHADS 2 score (p = 0.004, 0.035, and 0.002, respectively; Figure 3 ). Prominent features of patients with LA or LAA thrombus or sludge and CHADS 2 scores of 0 are listed in Table 2 .
| Age (yrs) | Gender | LVEF (%) | Other Condition | Sludge or Thrombus |
|---|---|---|---|---|
| 44 | M | 50 | Ventricular septal defect repair | Thrombus |
| 47 | M | 40 | Hypertrophic cardiomyopathy | Sludge |
| 53 | F | 50 | Postural orthostatic tachycardia | Thrombus |
| 60 | M | 55 | Mitral valve prolapse ∗ | Thrombus |
| 61 | F | 55 | Breast cancer | Thrombus |
| 72 | M | 45 | Severe atherosclerotic disease | Thrombus |
In the multivariate model, only 2 of 5 CHADS 2 score variables (congestive heart failure and hypertension) had statistically significant associations with the presence of thrombus or sludge in the LA and LAA ( Table 3 ). Diabetes mellitus and age >75 years did not increase the risk for thrombus and sludge formation in the LA or LAA. However, a history of heart failure significantly increased the risk for LA or LAA thrombus and sludge formation. Although this increased risk was observed with decreasing ejection fraction, it was most prominent in patients with heart failure who had LVEFs of ≤20%. An LVEF ≤20% was the strongest predictor of LA or LAA thrombus and sludge compared with the CHADS 2 score variables and higher ejection fractions ( Table 3 ).
| Variable | Odds Ratio | 95% Confidence Interval |
|---|---|---|
| History of congestive heart failure | 1.78 | 1.22–2.57 |
| LVEF ≤40% | 1.95 | 1.42–2.67 |
| LVEF ≤30% | 2.38 | 1.73–3.27 |
| LVEF ≤20% | 2.99 | 2.13–4.21 |
| Hypertension | 1.51 | 1.05–2.11 |
| Age >75 yrs | 0.92 | 0.63–1.23 |
| Diabetes mellitus | 1.18 | 0.84–1.63 |
| Previous stroke/TIA | 1.14 | 0.92–1.44 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


