Chapter 39 Diagnosis and Management of Asthma in Adults
Definition and Key Features of Asthma
Asthma is derived from the Greek word aazein, meaning “to labor in breathing” and was first used by Hippocrates, in 450 BCE, to describe a condition characterized by spasms of breathlessness. The present Global Initiative for Asthma (GINA) definition of the disease (Box 39-1) is a lengthy description of histopathologic, pathophysiologic, and clinical features that encompass the major disease characteristics. Fundamental features are airway hyperresponsiveness, chronic airway inflammation, disordered airway mucosal immunity, and structural changes to the airways (airway remodeling).
Box 39-1
Global Initiative for Asthma (GINA) Description of Asthma
From Global Strategy for Asthma Management and Prevention (Update 2009). Available at www.ginaaasthma.org.
Airway Hyperresponsiveness
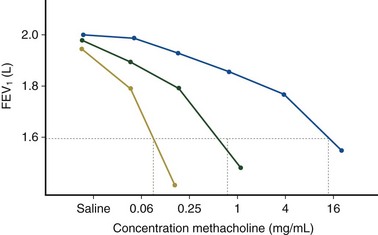
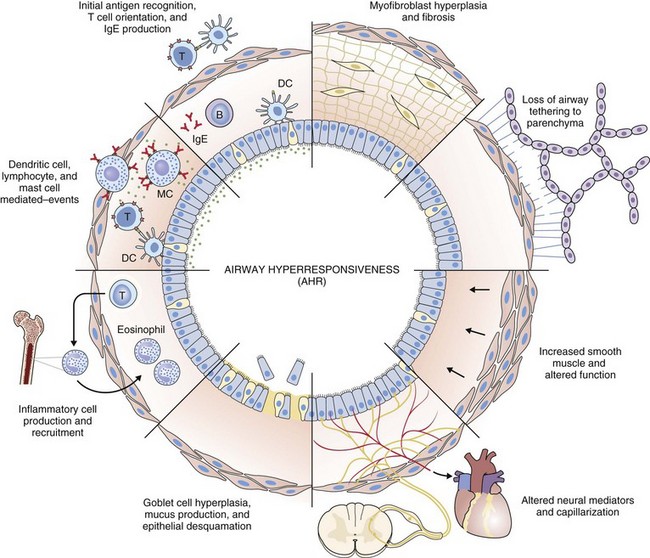
Airway hyperresponsiveness can be objectively demonstrated as a 20% fall in the forced expiratory volume in 1 second (FEV1) after inhalation of histamine or methacholine) at a concentration below 8 mg/mL. This represents an abnormal effector response of airway smooth muscle, characterized by heightened pharmacologic sensitivity and reactivity to the bronchoconstrictor stimulus (Figure 39-1). Naturally occurring airway hyperresponsiveness reflects an abnormally amplified response of airway nerves and mast cells to exogenous stimuli, as well as an intrinsic abnormality of the airway smooth muscle response. The basis for this generalized hyperresponsiveness is not entirely clear, but sensitization of airway nerves, mast cells, and smooth muscle by inflammatory mediators, along with loss of epithelial barrier function, reduced production of bronchoprotective factors, an intrinsic abnormality of airway smooth muscle, and structural changes to the airway, all are likely to play a part (Figure 39-2). One key pathologic feature associated with airway hyperresponsiveness in asthma not seen in nonasthmatic eosinophilic bronchitis is infiltration of the bronchial smooth muscle layer by mast cells, implying that the interaction between these cells is fundamentally important in the pathogenesis of airway hyperresponsiveness.
Chronic Airway Inflammation
Histopathologic examination of postmortem specimens from patients with fatal asthma show an inflammatory response characterized in many cases by the presence of airway eosinophilia. Typically, eosinophilic infiltration can be found throughout the airway wall, within thick viscid plugs that occlude the airway lumen and often extend into the lung parenchyma and alveolar spaces and even into adjacent blood vessels (see Figure 39-2). In addition, extensive eosinophilic degranulation with deposition of major basic proteins occurs. Associated findings include widespread shedding of the airway surface epithelium, thickening of the reticular basement membrane, and enlargement of airway smooth muscle and submucosal glands. A minority of patients dying from asthma, particularly those with sudden-onset fatal asthma, exhibit evidence of eosinophilic inflammation. In these cases it is believed that widespread mast cell degranulation is the primary event with evidence for a relative excess of neutrophils in the distal airways and parenchyma.
Structural Changes to the Airway
Structural changes in airway morphology (airway remodeling) probably occur as a result of chronic airway inflammation and dysfunction. These changes are likely to be the basis for the accelerated lung function decline and fixed airflow obstruction seen in some patients with asthma. Key features of airway remodeling include thickening of the subepithelial basement membrane caused by abnormal deposition of collagen, increased airway smooth muscle bulk, increased mucus-secreting cells, and increased airway vascularity (see Figure 39-2). In severe asthma, bronchiectasis, small airway fibrosis, and emphysema may be features. The bronchiectasis is associated with sensitization to Aspergillus.
Pathophysiologic Basis of Asthma
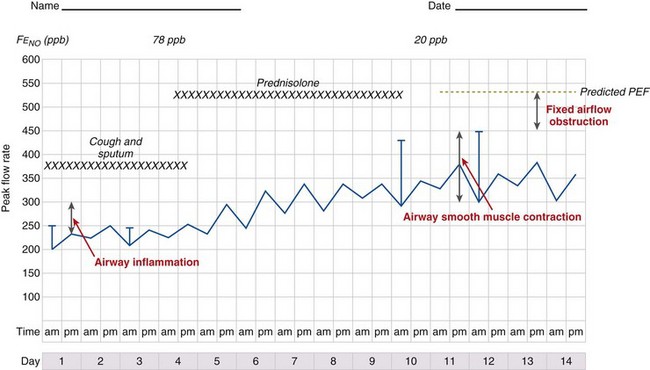
It is likely that these factors contribute to airflow obstruction and morbidity in different ways (Figure 39-3). Airway hyperresponsiveness is responsible for short-term, bronchodilator-responsive variable airflow obstruction which is the basis for many of the day-to-day symptoms experienced by patients and is suppressed by bronchodilator therapy, and to a lesser extent corticosteroids. Bronchitis, which may be eosinophilic and corticosteroid-responsive or neutrophilic and corticosteroid-unresponsive, manifests with cough and sputum and a relatively bronchodilator-resistant but potentially corticosteroid-responsive airflow obstruction of more gradual onset. Inflammation-mediated airflow obstruction is particularly important in the pathogenesis of asthma attacks. Cough and a small amount of sputum production is common in asthma. It probably reflects airway inflammation but also may be due to cough reflex hypersensitivity, a common but poorly understood and difficult-to-treat aspect of asthma and other airway diseases. Airway damage may manifest with disability caused by bronchodilator and corticosteroid-resistant airflow obstruction or mucus retention and infection as a result of airway and lung parenchymal damage. Extrapulmonary conditions linked to the inflammatory airway disease or independent of it also may contribute to symptoms (Box 39-2).
Diagnosis
One of the problems facing clinicians and epidemiologists is the absence of a “gold standard” modality for defining or diagnosing asthma. Characteristic clinical features (Box 39-3) coupled with objective demonstration of variable airflow obstruction and/or airway hyperresponsiveness usually will provide sufficient evidence to make the diagnosis.
Box 39-3
Factors Affecting Likelihood of Asthma as Cause of Respiratory Symptoms*
Factors Associated With Increased Probability of Asthma
More than one of the following symptoms: cough, breathlessness, wheeze, chest tightness
Symptoms worse at night and in the early morning
Symptoms occurring in response to exercise, allergen exposure, and cold air
Symptoms developing after taking aspirin or beta blockers
Family history of asthma and/or atopic disorder
Variable wheeze heard on auscultation of the chest
Variable PEF (see Clinical Examination Section)
Factors Associated With Lower Probability of Asthma
Prominent dizziness, light-headedness, peripheral tingling
Repeatedly normal physical examination of chest when symptomatic
Significant smoking history (i.e., more than 20 pack-years)
History
Asthma symptoms typically are variable reflecting variability in airflow obstruction and other aspects of the disease. Exaggerated diurnal variation in physiologic bronchomotor tone causes airflow obstruction and symptoms at night, maximally at 4 AM. Other symptoms reflect airway hyperresponsiveness to a multitude of possible trigger factors. Symptoms occurring after exercise, after exposure to allergens, and in response to inhaled noxious fumes such as cigarette smoke are most commonly reported. Box 39-3 lists some of the symptoms that increase and decrease the probability of asthma. An assessment of disease severity can be made from the frequency of daily symptoms, exercise limitation, nocturnal wakening, and exacerbation frequency (Table 39-1).
Clinical Examination
Measuring Variability in Airflow Obstruction
Diurnal variation in peak expiratory flow (PEF): Calculated from serial PEF measurements (see Figure 39-3); typically expressed as the difference between the highest and lowest daily readings as a percentage of the mean (see Table 39-1)
Assessment of the bronchodilator response: Measured as the change (improvement) in FEV1 or PEF from baseline with either a short-acting bronchodilator (bronchodilator responsiveness) or after a therapeutic trial of corticosteroid (steroid responsiveness)
Assessment of airway responsiveness: Measuring the fall in FEV1 in response to bronchoconstrictor stimuli, including pharmacologic agents (methacholine or histamine challenge tests), exercise, and allergen challenge (see Figure 39-1)
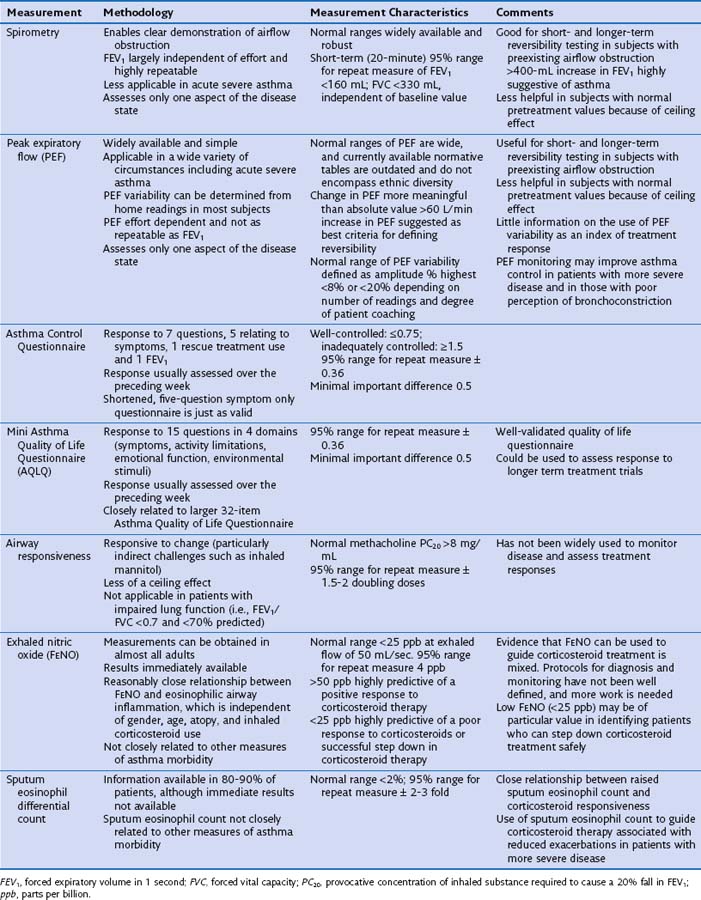
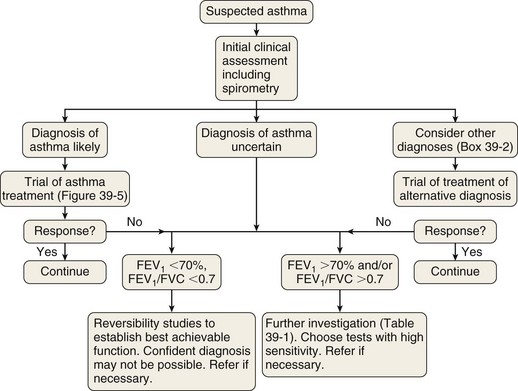
The measurement characteristics of the various tests to assess variable airflow obstruction and asthma in general are outlined in Table 39-1. An algorithm for the assessment of patients with suspected asthma is presented in Figure 39-4.
Diagnostic Pathway
The diagnostic pathway followed will depend on the pretest probability for the diagnosis, response to therapy, and level of clinical suspicion for an alternative diagnosis. The scope of the differential diagnosis is quite different in patients with and without airflow obstruction (see Box 39-2), and the main diagnostic question also is different. In the former, the clinician often is seeking support for a clinical diagnosis of asthma and the initiation of antiasthma therapy; in the latter, the evidence for an airway problem is more clear-cut, and the question is not whether inhalers should be used but how intensive the corticosteroid component of that therapy should be.