Chapter 6 Control of Ventilation and Respiratory Muscles
The Respiratory Centers
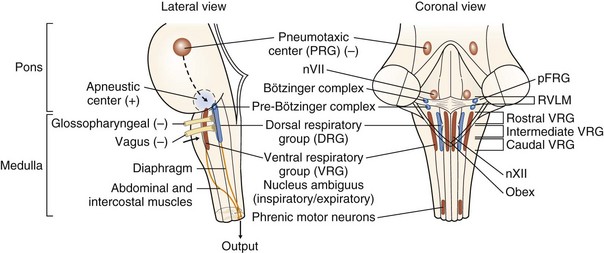
Early studies of the neural control of breathing involved the section and ablation of various brain stem structures. From these studies emerged the classical description of the neural control of breathing that required centers in the medulla for the rhythmic generation of ventilatory drive plus additional areas in the pons (traditionally known as the pneumotaxic and apneustic centers) that modulated and regulated the basic rhythm. Nowadays, the very complex and inadequately explored and understood respiratory center structure and function can be summarized as follows (Figure 6-1):
• Primary centers responsible for the generation of respiratory rhythm are located in the medulla. Within the medulla, there are two bilateral aggregations of neurons having respiratory related activity.
• An area of the ventrolateral medulla next to the nucleus ambiguus, the pre-Bötzinger complex, is hypothesized to be a critical site for respiratory rhythmogenesis. Current theory proposes that a group of pacemaker neurons depolarize, fire, and repolarize in a rhythmic fashion. This endogenous oscillatory activity can be modulated by afferent inputs, generating an efferent output that is translated into the respiratory drive. Apart from the pre-Bötzinger complex principally involved in controlling inspiratory motor activity, the retrotrapezoid-parafacial respiratory group (RTN/pFRG) appears to play at least a modulatory role and may be a conditional oscillator that controls active expiration.
• An additional mechanism is voluntary control of the respiratory muscles, signals for which originate in the motor cortex and pass directly to the spinal motor neurons by way of the corticospinal tracts. The medullary respiratory control center is bypassed. The voluntary control competes with automatic control at the level of the spinal motor neuron.
Afferent Inputs to the Respiratory Centers
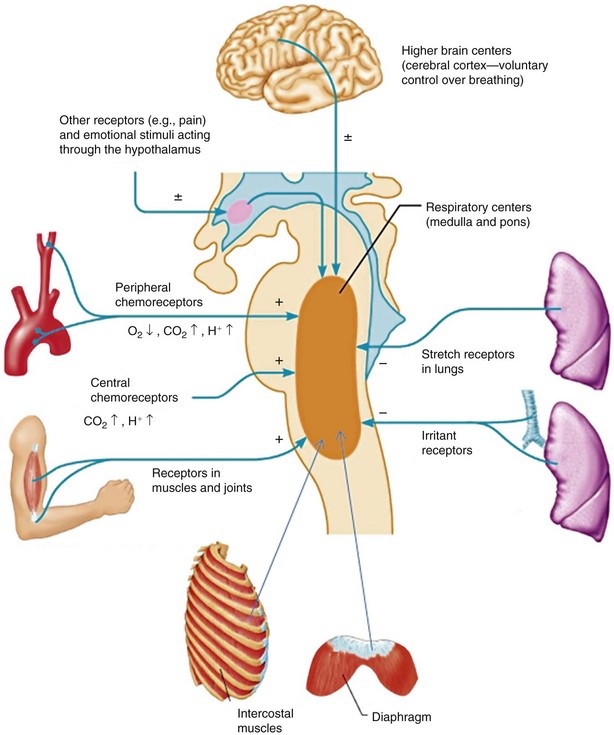
The respiratory controller receives information from a variety of sources. Some of these involve the relatively straightforward chemoreceptor signals that provide closed-loop information on the gas exchange functions of the lung. These signals arise mainly from the central and peripheral chemoreceptors that mediate the response to hypoxia, hypercapnia, and acidemia. In addition, at any given time, many other inputs from the upper airways, the lung, the respiratory muscles, and the thoracic cage may be important in determining ventilatory drive (Figure 6-2). The states of cortical arousal, sleep, and emotion play important roles in the level of ventilation and the response to other stimuli.
The Efferent Limb: the Respiratory Muscles
Functional Anatomy
The Intercostal Muscles
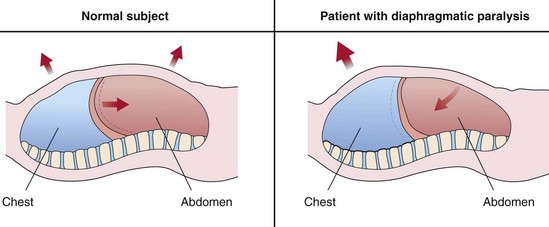
The external intercostal muscles have an inspiratory action on the rib cage, whereas the internal intercostal muscles are expiratory. An illustrative clinical example of the “isolated” inspiratory action of the intercostal muscles is offered by bilateral diaphragmatic paralysis. In patients with this deficit, inspiration is accomplished solely by the rib cage muscles. As a result, the rib cage expands during inspiration, and the pleural pressure falls. Because the diaphragm is flaccid and no transdiaphragmatic pressure can be developed, the fall in pleural pressure is transmitted to the abdomen, causing an equal fall in the abdominal pressure. Hence, the abdomen moves paradoxically inward during inspiration, opposing the inflation of the lung (Figure 6-3). This paradoxical motion is the cardinal sign of diaphragmatic paralysis on clinical examination and is invariably present in the supine posture, during which the abdominal muscles usually remain relaxed during the entire respiratory cycle. However, this sign may be absent in the erect posture.
The Diaphragm
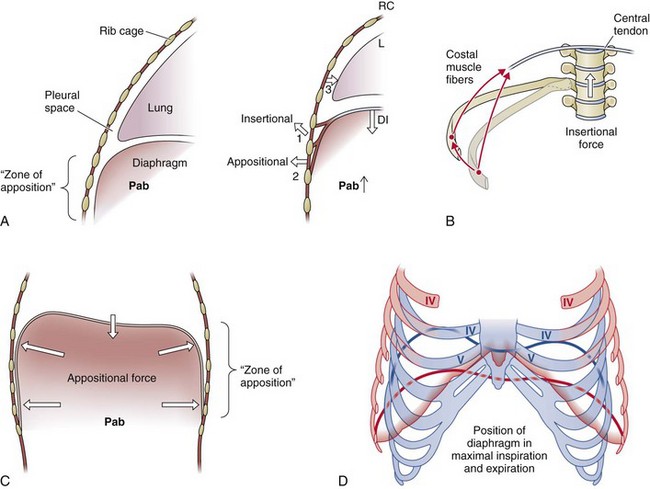
The shape of the relaxed diaphragm at the end of a normal expiration (at functional residual capacity [FRC]) is that of two domes joined by a “saddle” that runs from the sternum to the anterior surface of the spinal column (Figure 6-4). The motor innervation of the diaphragm is from the phrenic nerves, which also provide a proprioceptive supply to the muscle. When tension develops within the diaphragmatic muscle fibers, a caudally oriented force is applied on the central tendon, and the dome of the diaphragm descends; this descent has two effects. First, it expands the thoracic cavity along its craniocaudal axis, and consequently the pleural pressure falls. Second, it produces a caudal displacement of the abdominal visceral contents and an increase in the abdominal pressure, which in turn results in an outward motion of the ventral abdominal wall and the lower rib cage (appositional force). Thus, when the diaphragm contracts, a cranially oriented force is being applied by the costal diaphragmatic fibers to the upper margins of the lower six ribs that has the effect of lifting and rotating them outward (insertional force) (see Figure 6-4). The actions mediated by the changes in pleural and abdominal pressures are more complex. Viewed as the only muscle acting on the rib cage, the diaphragm has two opposing effects when it contracts: On the upper rib cage, it causes a decrease in the anteroposterior diameter, and this expiratory action results primarily from the fall in pleural pressure (see Figure 6-4). On the lower rib cage, it causes an expansion. In fact, this is the pattern of chest wall motion observed in tetraplegic patients with transection injury at the fifth cervical segment of the spinal cord or below, who have complete paralysis of the inspiratory muscles except for the diaphragm. This inspiratory action on the lower rib cage is caused by the concomitant action of two different forces, the “insertional” force already described and the “appositional” force.