Both hyperkalemia and hypokalemia may be related to heart failure (HF) therapy and are associated with adverse outcomes. Abnormalities in serum potassium levels in hospitalized patients with HF and reduced ejection fraction (EF) have not been previously investigated. A post hoc analysis was performed in 1,907 hospitalized patients with worsening HF and reduced EF in the placebo arm of the Efficacy of Vasopressin Antagonism in HF Outcome Study with Tolvaptan (EVEREST) trial. Serum potassium was measured at randomization and at discharge or day 7. The co-primary end points were all-cause mortality (ACM) and cardiovascular mortality or the first HF hospitalization (CVM + HFH). The association between inhospital change in potassium levels and time to outcomes was evaluated using multivariate Cox regression models. Study participants had a mean age of 65.6 ± 12.0 years and were on optimal guideline-directed medical therapies, including β blockers (77%), angiotensin-converting enzyme inhibitors/angiotensin receptor blockers (85%), and aldosterone antagonists (55%). Baseline potassium concentration was 4.3 ± 0.6 mEq/l, and hyperkalemia or hypokalemia was seen in 6.5% of the participants. On average, serum potassium level increased by 0.21 ± 0.66 mEq/l, p <0.0001, during hospitalization. Inhospital potassium change was not associated with either the primary or the secondary end point over a median follow-up of 9.9 months. In conclusion, in patients with reduced EF hospitalized for worsening HF, serum potassium abnormalities are common at baseline (within 48 hours of admission) and potassium levels increase during hospitalization, despite aggressive diuretic therapy. However, they are not associated with all-cause or CVM or HFH. Inhospital changes in potassium may limit the implementation of evidence-based therapies such as mineralocorticoid receptor antagonists.
Potassium is the major cation in the intracellular space and an important determinant of myocyte function. Abnormalities in potassium homeostasis are common in both inpatients and outpatients with heart failure (HF) and may be associated with worse outcomes. The inpatient setting is a vulnerable period for alterations in potassium homeostasis because of more intensive diuretic therapy and frequent fluctuations of renal function. Hypokalemia is frequently observed in patients with HF, which may be related to diuretic therapy or the neurohormonal activation. Diuretic therapy, especially during hospitalization for worsening HF, may heighten neurohormonal activation and may result in further disrupt potassium levels. Conversely, hyperkalemia is also frequent in patients with HF, related to both abnormal renal function and use of therapies to modulate RAAS in patients with reduced ejection fraction (EF). Similar to hypokalemia, even mild degrees of hyperkalemia (i.e., K >5 mEq/l) are associated with increased all-cause mortality risk. The Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study with Tolvaptan (EVEREST) trial provides a unique opportunity to perform an in-depth characterization of admission serum potassium levels and changes during hospitalization in a large contemporary cohort of patients hospitalized for worsening HF with reduced EF, who were well treated with evidence-based therapies. Specifically, the objectives of this post hoc analysis were to (1) determine the baseline prevalence of potassium abnormalities; (2) describe the changes in potassium during hospitalization; and (3) assess the prognostic value of baseline serum potassium and of inhospital potassium level changes, on postdischarge morbidity and mortality.
Methods
The study design and primary results of the EVEREST trial have been previously reported. Briefly, EVEREST was a global, prospective, randomized, double-blind, placebo-controlled trial designed to examine the short- and long-term efficacy and safety of tolvaptan, a vasopressin-2 receptor antagonist. Patients aged ≥18 years, a left ventricular EF ≤40% hospitalized for worsening HF, and presenting with 2 or more signs or symptoms of volume overload were eligible for enrollment. Relevant exclusion criteria included serum creatinine >3.5 mg/dl, serum potassium level >5.5 mEq/l, or a life expectancy of <6 months. The ethics committee and institutional review board of each participating site approved the study protocol. After providing informed consent, study participants were randomized to receive oral tolvaptan at a 30-mg fixed dose or matching placebo within 48 hours of hospital admission, which was continued for at least 60 days. Background medical therapy for HF was left to the discretion of the treating physician, but guideline-based recommendations for optimal medical management were included in the study protocol.
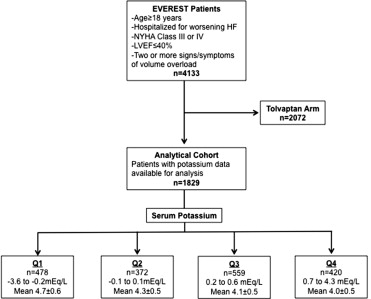
Because tolvaptan may influence potassium levels at discharge or day 7, this analysis was restricted to the placebo cohort. Serum potassium level (mg/dl) was measured at enrollment (within 48 hours after admission and screening potassium level) and at discharge or day 7, whichever occurred first. All laboratory testing was performed at 5 central facilities and laboratory assay variability was cross validated between sites to verify result consistency. Inhospital potassium change was calculated by subtracting the baseline potassium level from discharge or day 7 and was the primary predictor in this post hoc analysis (expressed as a continuous function per 1 mg/dl increase). No nonlinear effects were detected, and therefore, no transformation of data was undertaken. For descriptive purposes, subjects were divided by quartiles of inhospital potassium change. The overall study design and final analytical cohort selection are displayed in Figure 1 .
An independent blinded adjudication committee determined the specific causes of death and reasons for rehospitalization. The 2 co-primary end points in this retrospective post hoc analysis were all-cause mortality (ACM) and a composite of cardiovascular mortality (CVM) or heart failure hospitalization (HHF), the same as in the overall EVEREST trial. Secondary end points included death from other causes, rehospitalization, worsening HF (defined as death, hospitalization, or unplanned office visit for HF), and combined CV mortality and rehospitalization (including hospitalization for HF, acute myocardial infarction, arrhythmia, and stroke). Other CV hospitalization included unstable angina and pulmonary embolism.
Baseline demographic characteristics, clinical history, signs and symptoms of HF, vital signs, laboratory parameters, and admission and discharge medications were compared across quartiles of inhospital potassium changes. The median follow-up was 9.9 (interquartile range 5.3 to 16.1) months. Continuous variables were expressed as mean ± SD if normally distributed and median (interquartile range) if not normally distributed. Categorical variables were expressed as number (percentage). p Values for trend in all 4 quartiles are provided for baseline demographics and clinical outcomes. Outcomes were assessed as time to the first event using Cox proportional hazard models. Kaplan–Meier curves by inhospital serum potassium changes were constructed for both primary end points and compared using log-rank tests. The proportional hazards assumption (by Kolmogorov-type supremum tests) was upheld for both primary end points. Effect sizes were reported as hazard ratios (HRs) with 95% confidence intervals (CIs). Multivariate models included 23 prespecified covariates, including demographic characteristics (gender and region of origin), clinical characteristics (ischemic HF origin, atrial fibrillation on admission electrocardiogram, coronary artery disease, diabetes, hypertension, chronic obstructive pulmonary disease, chronic kidney disease [CKD, defined as an estimated glomerular filtrate rate [GFR] <60 ml/min/1.73 m 2 by the Modification of Diet in Renal Disease formula], New York Heart Association [NYHA] class IV symptoms, supine systolic blood pressure [SBP], and laboratory and diagnostic testing [QRS duration on admission electrocardiogram, EF, serum sodium, blood urea nitrogen, and B-type natriuretic peptide {BNP} on admission]), and baseline medication use (angiotensin-converting enzyme inhibitors [ACEI] or angiotensin II receptor blockers [ARB], β blockers, mineralocorticoid receptor antagonists [MRA], digoxin, and intravenous inotropes). No evidence of significant collinearity between inhospital potassium changes and the covariate set was detected. Spearman correlation coefficients were determined for the correlation between inhospital potassium changes and age, inhospital changes in weight, SBP, GFR, and BNP. All statistical analyses were performed using SAS version 9.2 (S.A.S Institute Inc., Cary, North Carolina).
Results
Overall, from October 7, 2003, to February 3, 2006, 2,061 patients were assigned to the placebo arm of the EVEREST trial. Of those, 1,907 (73%) had serum potassium measured at baseline and at discharge or day 7 and were included in the analysis. Serum potassium level at baseline and inhospital potassium change were normally distributed ( Figure 2 ). Seventy-five patients (3.9%) had a potassium level at baseline of <3.4 mEq/l and 51 patients (2.7%) had a potassium level at baseline of >5.4 mEq/l. Overall, 279 patients (14.6%) developed hyperkalemia (K >5 mEq/l) at discharge. Table 1 details the baseline characteristics across quartiles of inhospital potassium changes. Of note, 23% of patients experienced an increase in potassium levels from 0.7 to 4.3 mEq/l during hospitalization. Baseline supine SBP and heart rate and prevalence of NYHA class IV symptoms were higher, whereas serum albumin was lower in patients in the highest quartile of potassium increase (all p <0.04). Patients with potassium increases during hospitalization tended to have lower creatinine and higher GFR and were more likely to be on ACEI-ARBs (88%) and MRAs (70%) at discharge (all p <0.02). Of note, no differences were observed in mean EF, ischemic origin of HF, and signs and symptoms of HF, across quartiles of inhospital potassium change. There were only weak correlations between inhospital changes in potassium levels and inhospital changes in weight ( r = −0.015, p = 0.53), SBP ( r = −0.07, p = 0.01), BNP ( r = −0.07, p = 0.01), and GFR ( r = −0.18, p <0.001).
| Variable | Quartiles of Serum Potassium Change (mEq/L) | ||||
|---|---|---|---|---|---|
| Q1 (-3.6 to -0.2) | Q2 (-0.1 to 0.1) | Q3 (0.2 to 0.6) | Q4 (0.7 to 4.3) | P-Value ∗ | |
| n = 478 | n = 372 | n = 559 | n = 420 | ||
| Age (years) | 66.6±11.8 | 65.4±12.4 | 65.2±11.9 | 64.4±12.0 | 0.051 |
| Men | 380 (77.7%) | 303 (75.9%) | 431 (74.3%) | 330 (75.2%) | 0.623 |
| Ejection fraction (%) | 27.1±8.1 | 27.6±8.1 | 28.1±8.2 | 27.4±8.9 | 0.260 |
| Dyspnea | 442 (92.7%) | 365 (92.9%) | 512 (90.1) | 389 (89.2) | 0.137 |
| Jugular venous distension | 131 (27.5%) | 102 (26.2%) | 147 (26.1%) | 119 (27.7%) | 0.911 |
| Supine Systolic Blood Pressure (mm Hg) | 118.9±19 | 118.5±19.1 | 121.3±19 | 122.3±20.4 | 0.007 |
| New York Heart Association Class IV | 177 (36.2%) | 167 (42%) | 217 (37.4%) | 194 (44.2%) | 0.040 |
| Atrial Fibrillation, | 145 (29.7%) | 108 (27.1%) | 160 (27.6%) | 134(30.6%) | 0.618 |
| Ischemic Heart Failure | 317 (65.9%) | 250 (63.1%) | 368 (64%) | 305 (70.4%) | 0.101 |
| Hypertension | 348 (71.2%) | 271 (67.9%) | 420 (72.4%) | 314 (71.5%) | 0.482 |
| Coronary Artery Disease | 338 (69.3%) | 279 (70.1%) | 404 (69.7%) | 319 (72.7%) | 0.671 |
| Renal Disease | 157 (31.1%) | 119 (29.8%) | 140 (24.1%) | 96 (21.9%) | 0.001 |
| Diabetes | 190 (38.9%) | 155 (38.8%) | 206 (35.5%) | 165 (37.6%) | 0.645 |
| Weight (kg) | 83.2±18.9 | 83.3±18.4 | 83.7±17.7 | 82.7±19.4 | 0.879 |
| Heart rate (bpm) | 79±15 | 78±16 | 79±15 | 82±16 | 0.020 |
| Albumin, mg/dL | 3.8±0.5 | 3.8±0.5 | 3.8±0.5 | 3.7±0.5 | 0.015 |
| B-type natriuretic peptide (pg/mL), (IQR) | 1420±3989 | 1633±6132 | 1009±1234 | 1495±4428 | 0.195 |
| Blood urea nitrogen (mg/dL) | 32.4±16.9 | 30.9±18.1 | 29.5±16.9 | 27.4±13.6 ‡ | <0.001 |
| Creatinine (mg/dL) | 1.4 ±0.5 | 1.4±0.5 | 1.4±0.7 | 1.3±0.4 ‡ | 0.001 |
| Estimated Glomerular Filtration Rate (mL/min) | 52.8±19.7 | 56.3±22.3 ∗ | 56.7±20.9 | 59.0±20.7 | <0.001 |
| Medications at admission | |||||
| Angiotensin Converting Enzyme Inhibitors/Angiotensin Receptor Blockers | 413 (84.6%) | 334 (83.9%) | 490 (84.9%) | 375 (85.4%) | 0.944 |
| Beta Blockers | 345 (70.7%) | 289 (72.6%) | 394 (68.3%) | 316 (72%) | 0.445 |
| DIgoxin | 241 (49.4%) | 183 (46%) | 279 (48.4%) | 221 (50.3%) | 0.623 |
| Mineralacorticoid Receptor Antagonists | 254 (52%) | 225 (56.5%) | 320 (55.5%) | 266 (60.6%) | 0.073 |
| Medications at discharge | |||||
| Angiotensin Converting Enzyme Inhibitors/Angiotensin Receptor Blockers | 390 (80.7%) | 332 (84.7%) | 499 (86.5%) | 374 (87.6%) | 0.018 |
| Beta Blockers | 350 (72.5%) | 291 (74.2%) | 421 (73%) | 338 (79.2%) | 0.084 |
| Digoxin | 232 (48%) | 185 (47.2%) | 274 (47.5%) | 209 (48.9%) | 0.958 |
| Mineralacorticoid Receptor Antagonists | 274 (56.7%) | 232 (59.2%) | 365 (63.3%) | 299 (70%) | <0.001 |
∗ P < 0.05 Q2 vs Q1. † P < 0.05 Q3 vs Q2. ‡ P < 0.05 Q4 vs Q3.
During a median follow-up of 9.9 months, there were 475 patients (24.9%) who died of which only 32 occurred during hospitalization. There were 363 patients (19%) who had CVM, 523 (27.4%) had an HFH, and 672 (35.2%) had worsening HF. There were no significant differences in any of the outcomes across the quartiles of inhospital potassium change; however, a trend toward decreased event rates was observed in patients experiencing the greatest serum potassium increase although it did not reach statistical significance (p = 0.075; Table 2 ). Other CV hospitalization, not including HF, MI, arrhythmia, or stroke, was lowest in the patients with greatest serum potassium increase during hospitalization (p <0.001). Time to the first event was also not significantly different by Kaplan–Meier method across quartiles of inhospital change for ACM and CVM or HFH ( Figures 3 and 4 ). In univariate analysis, every 1-mEq/l increase in serum potassium was predictive neither of ACM (HR 1.01; 95% CI 0.874 to 1.17, p = 0.89) nor of the composite end point (HR 0.99; 95% CI 0.88 to 1.10, p = 0.81) with similar results in multivariate analyses with p = 0.08 and 0.66, respectively.



