Chapter 7 Central nervous system control of cardiovascular function
After reading this chapter, you should:
The brain exerts powerful effects on cardiovascular behaviour, predominantly by altering sympathetic motor drive to the heart and to vascular smooth muscle. While you may read about sympathetic activation specifically in relation to cardiac function or arteriolar resistance or venous capacitance, depending on the topic being discussed, the truth is that centrally mediated sympathetic activation almost always involves increased drive to both heart and blood vessels and in non-selective increases in vasomotor drive to arteries and veins. Therefore, heart rate and blood pressure usually change in parallel. This parallelism is convenient because it means that changes in any of these parameters can be used as an (approximate) index of sympathetic activation. In practice, the parameter used routinely is the change in heart rate, because ECGs can be recorded continually in moving subjects so much more easily than blood pressure.
ORGANIZATION OF CENTRAL CARDIOVASCULAR CONTROL
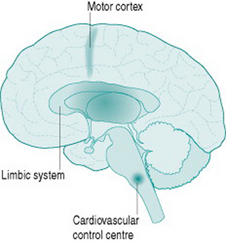
Although sympathetic outflow to the cardiovascular system can be affected by pathways arising in many areas of the brain, there are for our purposes three primary sources of this activation (Fig. 7.1). The first is the cardiovascular control centre lying in the medulla of the hindbrain, which is the final common pathway for all descending sympathetic information. This hindbrain centre receives reflex inputs directly from peripheral sensory nerves, but also receives descending inputs from two areas of the forebrain. The most important of these are the limbic system, which includes the hypothalamus and several neighbouring nuclei, mediates emotional arousal and determines levels of consciousness, and the motor cortex from which all instructions for muscle movement must originate.
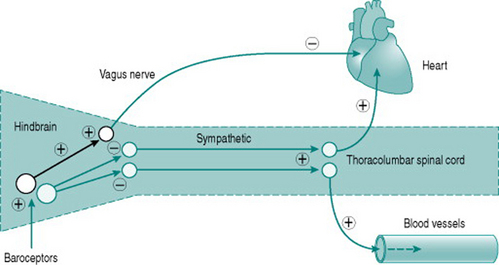
Neuroscientists are still unravelling the specific neural connections and neurotransmitters involved in mediation of hindbrain regulation of the cardiovascular system. However, we can regard the cardiovascular control centre as consisting of two sets of tonically active descending neurons, one producing continual activation of the sympathetic outflows to heart and blood vessels and one producing continual inhibition of heart rate via the vagus (Fig. 7.2). As a result of this ongoing activity, there is usually some degree of vasoconstrictor tone in both arterial and venous vessels, while the heart rate at rest reflects the algebraic sum of inputs to the sinoatrial node of both sympathetic and vagal activity.
LOW-PRESSURE AND HIGH-PRESSURE BAROREFLEXES
Baroreflex effects on sympathetic cardiovascular control
The moment-to-moment regulation of blood pressure is via reflexes that operate through inputs to the hindbrain centre from mechanosensitive sensory axons monitoring pressure in the aorta and the carotid arteries (high-pressure baroreceptors) and in the atria (low-pressure baroreceptors). In all cases, increased distension results in increased sensory firing and the sensory neurons inhibit activity of the hindbrain centre sympathetic neurons. In consequence, either increased blood pressure or increased central blood volume will reduce peripheral resistance, while falls in blood pressure or central blood volume have the opposite effects.
Low-pressure baroreflexes have a distinct pattern of cardiac and vascular responses, with reflex falls in peripheral resistance caused by increased atrial filling being usually accompanied by tachycardia. However, increases in atrial volume that raise stroke volume will also induce an arterial baroreflex and bradycardia. This means that heart rate changes may be an uncertain index of baroreflex activity since their magnitude will vary depending on the algebraic sum of low-pressure and high-pressure receptor inputs.
Baroreflex effects on hormonal cardiovascular control
Activation of low-pressure baroreceptors by atrial distension sends inhibitory inputs to the hypothalamus that depress release of vasopressin (antidiuretic hormone, ADH) from the posterior pituitary gland. As a result, less water is reabsorbed from the renal collecting ducts and plasma volume is reduced. Conversely, reduced atrial baroreceptor activity due to lower atrial pressure will increase vasopressin release, retaining water and elevating plasma volume.