A. 1.33
B. 1.50
C. 1.75
D. 2.00
E. 2.25
20. Refer to the data in Table 4.1. If the calculated pulmonary flow index is 5.0 L/min/m2, what is the pulmonary arteriolar resistance?
A.2.5 units m2
B.5.0 units m2
C.400 dynes –s cm−5
D.10.0 units m2
E. Cannot be determined from the available data
21. The patient then receives 100% oxygen therapy during the procedure.
The data in Table 4.2 are obtained. What is the Qp/Qs now?
Table 4.2 Cardiac Catheterization Data on 100% Inspired Oxygen

A. 5.0
B. 4.2
C. 3.7
D. 3.1
E. 2.9
22. In calculating the oxygen content during these conditions, the systemic arterial pO2 needs to be considered. What constant is multiplied by the pO2 to calculate O2 content?
A. 1.36
B. 3.14
C. 0.006
D. 0.03
E. 0.2
23. Which statement is correct regarding balloon pulmonary valvuloplasty?
A. Applicable to patients of all ages except newborns
B. Doppler maximum instantaneous gradient = 35 mm Hg with RVH is an indication for intervention
C. Placement of the exchange guidewire is preferred in the RPA
D. Optimal balloon diameter should be 1.5 times the size of the valve annulus
E. High pressure balloons (>10 atmospheres) are preferred in children
24. Which statement is correct regarding balloon atrial septostomy (BAS)?
A. Can be performed effectively in infants up to 3 months of age
B. Commonly indicated in PA with IVS
C. Appropriate inflation pressure = 5 atmospheres
D. A sign of success is mobility of septum primum tissue by 2D echo
E. Balloon should be pulled slowly and steadily across the atrial septum
25. When counseling the parents of a neonate who is about to undergo aortic balloon valvuloplasty, which of the following statement is true?
A. Freedom from need for repeat balloon valvuloplasty is 30%
B. Freedom from aortic valve replacement within the next 5 years is 30%
C. Freedom from death or heart transplantation within the next 10 years is >80%
D. If the post-valvuloplasty gradient is >25 mm Hg, need for re-intervention is rare
E. Best results are attained in patients with LVEF <10%
26. In which patient is there an ACC/AHA class 1 indication for catheter-based intervention?
A. 20-year-old patient, asymptomatic, pulmonary valve mean Doppler gradient = 24 mm Hg
B. 30-year-old patient with refractory migraines and a PFO
C. 40-year-old patient, asymptomatic, pulmonary valve catheter gradient = 54 mm Hg
D. 25-year-old patient, asymptomatic, with a continuous murmur and small PDA on echo
E. 28-year-old patient with a small membranous VSD and recurrent endocarditis
27. During the pre-cath echo for potential ASD device closure, which rim needs particular attention? Complete absence of this portion of the septum may make stabilization of currently available devices difficult.
A. Anterior–superior
B. Posterior–superior
C. Anterior-retroaortic
D. Posterior–inferior
E. Atrio-ventricular
28. Which complication is least common after cardiac catheterization?
A. Low-grade fever for 4 to 8 hours
B. Hematoma at the catheterization site
C. Transient (<48 hours) occlusion of the catheterized vessel
D. Transient arrhythmias
E. Dye allergy
29. Conduction abnormalities following ASD device closure typically:
A. Resolve within the first 24 hours after device placement
B. Resolve 2 weeks after device placement
C.Progress into complete heart block in >15% of patients
D. Resolve only if aspirin therapy is continued for 1 year
E. Persist causing extra-nodal pathways to develop
30. To dilate an aortic valve with a 10-mm annulus, using a single-balloon technique, which size balloon is initially recommended?
A. 6 mm
B. 9 mm
C. 10 mm
D. 12 mm
E. 14 mm
31. To dilate a pulmonary valve with a-10 mm annulus, using a single-balloon technique, which size balloon is recommended?
A. 8 mm
B. 9 mm
C. 10 mm
D. 12 mm
E. 15 mm
32. The method used to reduce cardiac output during aortic valvuloplasty?
A. Rapid RV pacing
B. Slow RV pacing
C. Rapid LV pacing
D. Slow LV pacing
E. Dual-chamber pacing
33. Allergy to which metal has been reported following implantation of Amplatzer septal occluders?
A. Titanium
B. Selenium
C. Copper
D. Stainless steel
E. Nickel
34. The estimated incidence of erosion after septal occlusion device placement is closest to?
A. 1 in 1, 000, 000
B. 1 in 100, 000
C. 1 in 10, 000
D. 1 in 1000
E. 1 in 100
35. Risk of recoarctation in an infant undergoing balloon angioplasty for native coarctation of the aorta is?
A. 10% to 15%
B. 30% to 35%
C. 35% to 40%
D. 60% to 65%
E. 90%
36. A 17-year-old female is considered for device closure of a secundum ASD with a measured diameter of 26 mm by TTE. When discussing the advantages and disadvantages of using a septal occluder, which of the following is true?
A. The defect is too large for commercially available devices in the US
B. Risk of mortality is less than 1%
C. Antiplatelet therapy is needed for a minimum of 2 years post-procedure
D. Device embolization can only be treated with a surgical intervention
E. Risk of arrhythmia 3 months after the procedure is 30%
37. In a 6-year-old patient who undergoes intravascular stent placement, which lesion portends the greatest risk of restenosis?
A. Systemic vein stenosis
B. Branch pulmonary artery stenosis
C. Pulmonary vein stenosis
D. Coarctation of the aorta
E. Systemic arterial branch stenosis
38. As an interventional pediatric cardiologist, you are meeting with the family of a 1-week-old child with HLHS who will be undergoing stage I of the hybrid procedure. They ask what your role will be in the procedure. You answer that your primary role will be which of the following?
A. Balloon dilation of the pulmonary arteries
B. Stent placement in the coarctation
C. Atrial septectomy
D. Placement of a PDA stent
E. Aortic balloon valvuloplasty
39. A 16-year-old with cyanotic congenital heart disease is being considered for diagnostic cardiac catheterization, which of the following should be considered in the pre-procedure management of the patient?
A. May have clear liquids until 1 hour before the procedure
B. Should be admitted for intravenous hydration the evening before the procedure if the hemoglobin exceeds 19 gm/dL
C. Should receive warfarin therapy after the procedure
D. Should receive intravenous antibiotics 1 hour before the procedure
E. Should anticipate placement of a temporary intravenous pacemaker
40. A full-term infant with complete TGA and a pH of 7.1 is in the cath lab for BAS. After vascular access is obtained, which of the following is the best management of this situation?
A. BAS should be performed promptly
B. Obtain mixed venous saturation in the RA
C. Perform RV angiography to evaluate coronary anatomy
D. Perform RA angiography to assess right-to-left shunt
E. The procedure is not required when patients have a co-existent VSD
41. A 2-year-old patient is about to have balloon valvuloplasty for pulmonary stenosis. Which single balloon diameter to valve annulus ratio is appropriate?
A. 1:1.25
B. 0.8:1
C. 1:1
D. 1.5:1
E. 1.2:1
42. During a cardiac catheterization the following oxygen saturations are obtained:
SVC – 68%, RA – 69%, RV – 85%, MPA – 86%, Aorta – 99%
What is the likely lesion?
A. Supracardiac total anomalous pulmonary veins
B. Infracardiac total anomalous pulmonary veins
C. Sinus venosus ASD with partial anomalous pulmonary veins
D. Large PDA in a patient with severe pulmonary regurgitation
E. Pulmonary A-V fistula
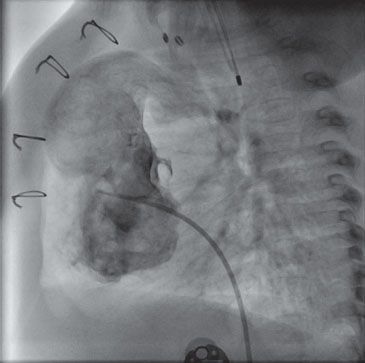
43. In a 6-month-old child, the angiogram in Figure 4.1. was likely performed because which of the following?
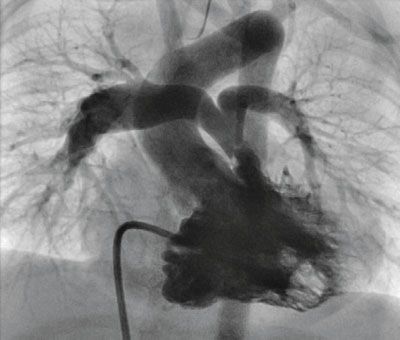
A. Transthoracic echocardiography could not visualize the proximal LPA
B. In this lesion calculation of pulmonary vascular resistance (PVR) is essential before surgical repair
C. Pulmonary balloon valvuloplasty was subsequently performed
D. Device closure of the ASD is needed
E.Qp/Qs needs to be calculated prior to repair
44. A 22-year-old with tetralogy of Fallot (TOF) had a complete repair at age 5 years. The native pulmonary valve was initially preserved, but subsequently (age 14 years) he developed significant valvular stenosis and regurgitation, and the valve was replaced with a 27-mm porcine bioprosthesis. That valve has now developed severe regurgitation, and he undergoes cardiac catheterization with the plan to insert a transcatheter valve within the pulmonary bioprosthesis. The data in Table 4.3 are obtained during the study.
Table 4.3 Cardiac Catheterization Data

No residual shunts were identified during the study.
The data presented above are most consistent with which of the following?
A. Mild pulmonary regurgitation
B. Moderate pulmonary valve stenosis
C. Restrictive physiology
D. Cardiac tamponade
E. Severe tricuspid regurgitation
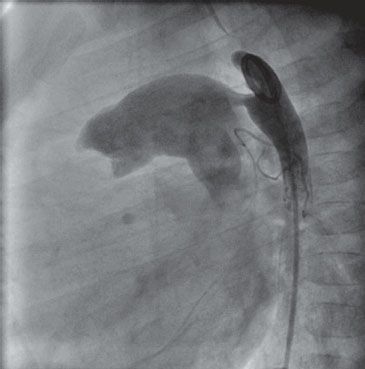
45. A 16-year-old patient from Africa with the chest radiograph in Figure 4.2. would be expected to have which of the following pressures if he underwent cardiac catheterization?
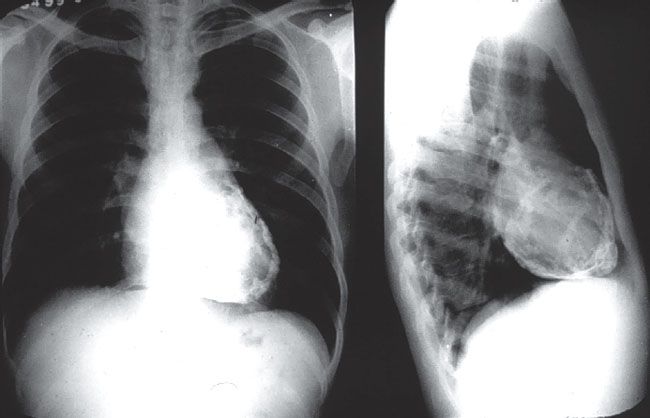
A. PCWP 6 mm Hg
B. Mean RAP 10 mm Hg
C. RVEDP 8 mm Hg
D. LVEDP 22 mm Hg
E. Ascending aorta 152/84 mm Hg
46. What is the course for catheter A in Figure 4.3.?
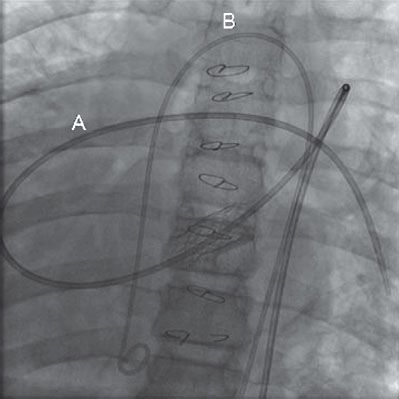
A. IVC, RA, RV, MPA, LPA
B. IVC, LA, RV, MPA, LPA
C. Azygos, SVC, RA. RV, MPA, LPA
D. IVC, RA, RV, RA
E. Azygos, RA, LA, LV, aorta
47. Which vessel is injected in the angiograph in Figure 4.4.?
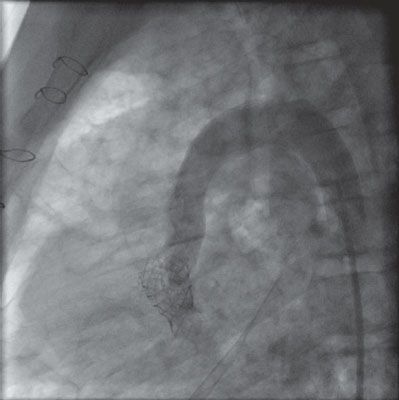
A. Inferior vena cava
B. Left pulmonary artery
C. Right pulmonary artery
D. Descending aorta
E. Azygos vein
48. This patient (Fig. 4.5) likely has which syndrome?
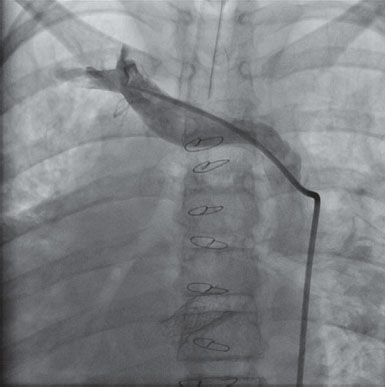
A. Kartagener
B. Turner
C. Noonan
D. Marfan
E. Down
49. After vascular access is obtained in a 10-year-old patient undergoing device closure of an ASD, which of following medications should be administered next?
A. Low molecular weight heparin 1 unit/kg
B. Intravenous heparin 100 units/kg
C. Aspirin 5 mg/kg
D. Clopidogrel 1 mg/kg
E. Gentamicin 50 mg/kg
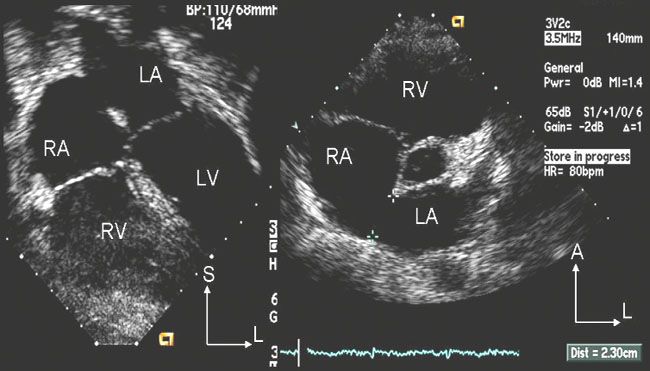
50. In an asymptomatic 10 kg, 2-year-old the transthoracic echocardiographic images in Figure 4.6. were obtained.
You are counseling the parents regarding type and timing of intervention. What would you say on the basis of these images?
A. This is a sinus venosus defect and therefore not amenable to device closure
B. The defect is well-centered and should be easily closed with a device
C. The defect is small and does not require closure at this time
D. The defect is large relative to the size of the child and some rims are deficient
E. The anterior/superior rim is very large, therefore suitable for device closure
51. The angiogram in Figure 4.7. was obtained in an 8-year-old patient.
Which syndrome does this patient have?
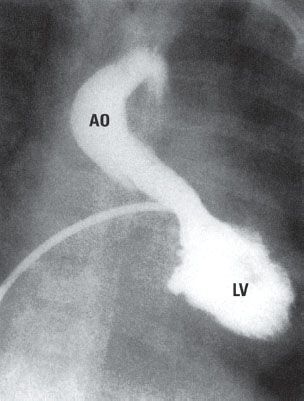
A. Marfan
B. Loeys–Dietz
C. Williams
D. Down
E. Ellis–van Creveld
52. Which of the following is true regarding creation of an atrial septal defect with catheter-based methods?
A. Considered in patients with severe PHTN refractory to medical management
B. Before performing blade septostomy, BAS should be attempted first
C. Vascular injury is a common complication of BAS
D. Of the four catheters available for atrial septostomy, the Rashkind is the only one with an end hole allowing placement over a guidewire and confirmation by contrast injection in the LA
E. Proper performance of a balloon septostomy includes pulling the catheter from the left atrium into the right atrium with slow steady pressure
53. In which form of congenital heart disease would ductal stenting be most appropriate?
A. Pulmonary atresia with IVS after RV outflow tract dilation
B. Pulmonary atresia with IVS and without RV outflow tract dilation
C. Tricuspid atresia with critical PS and a relatively straight duct
D. Critical pulmonary stenosis
E. Complex cyanotic CHD with proximal pulmonary artery stenosis
54. You are assessing a 5-year-old female for possible PDA closure. Her echo shows a large PDA with bidirectional shunting. There is no murmur on examination. Her echo is otherwise normal with no tricuspid regurgitation and no chamber enlargement. Her parents report she is a quiet child but does not seem to be limited in any way. How do you counsel her parents?
A. She would be best served by surgical closure of her PDA
B. She should proceed immediately for trans-catheter closure of her PDA
C. She should return in 3 years for follow-up echo evaluation
D. She should return in 6 to 12 months for follow-up echo
E. Invasive testing is needed before devising a plan for this child
55. A 14-month-old 11-kg child with newly diagnosed coarctation of the aorta is referred for further evaluation. He has a discrete juxtaductal coarctation with a mean gradient of 35 mm Hg across the narrowed segment. His LVEF = 65% and has mild-to-moderate LVH, and he has no other cardiac defects other than a bicuspid aortic valve with no obstruction. Right arm BP = 110/50 mm Hg, right leg = 75/45 mm Hg. His parents want to know if coarctation can be fixed without surgery. Which of the following would be the most appropriate answer to their question?
A. No intervention is needed at present and patient can be followed annually
B. Surgery is the best option and catheter intervention is contra-indicated
C. Surgery and catheter intervention are both options for the patient
D. Catheter intervention has a 50% risk of aortic dissection
E. Catheter intervention with stent implantation should be performed
56. Which of the following patients is most suitable for balloon angioplasty?
A. 2-month-old patient with coarctation, right arm BP = 100/60 mm Hg and right leg BP = 70/45 mm Hg
B. 12-year-old patient with Turner syndrome and recurrent coarctation; systolic coarctation gradient = 20 mm Hg
C. 18-year-old patient with tricuspid atresia, s/p Fontan and coarctation repair with systolic coarctation gradient ~15 mm Hg and mild LV dysfunction
D. 18-year-old patient with history of coarctation repair and now with systolic coarctation gradient ~10 mm Hg
E. 4-day-old patient with coarctation and no palpable femoral pulses
57. A 16-year-old patient with a history of coarctation of the aorta, status post end-to-end anastomosis at 1.5 years of age presents with hypertension and headaches. Right arm systolic BP = 136 mm Hg and leg BP = 118 mm Hg. A murmur is noted over his mid-left back. Which transcatheter systolic gradient would qualify as a Class I indication for balloon angioplasty?
A. 10 mm Hg
B. 15 mm Hg
C. 20 mm Hg
D. 30 mm Hg
E. 35 mm Hg
58. What other noncardiac test is indicated during your work up?
A. CBC
B. CT or MRI of head
C. Neurology consult
D. Electrolytes
E. Urinalysis
59. A newborn with an in utero diagnosis of PA-IVS is admitted to the NICU. An echocardiogram confirms the diagnosis. The infundibulum appears unobstructed. There is a mild pulmonary valve hypoplasia with no systolic or diastolic flow across the annulus. The RV is small with systolic pressure. The tricuspid valve is hypoplastic with a Z score = −2.5 with moderate regurgitation. The left ventricle has normal size and function. The coronary arteries were not well identified. A large ASD with right-to-left shunting is noted. The patient has a cardiac catheterization to perform balloon valvuloplasty. The angiograph in Figure 4.8. was obtained. What should be the next step for this patient?
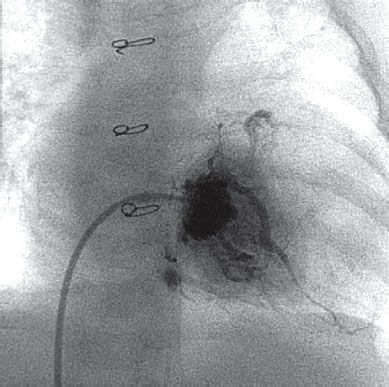
A. Dilation of the right coronary artery
B. BAS
C. Abort balloon valvuloplasty
D. Refer for surgical pulmonary valvotomy
E. Perform pulmonary valvuloplasty with MPA stent placement
60. A 17-year-old patient with no significant past medical history presents with a transient ischemic attack. She had a similar episode 2 years prior but failed to report that event. During the workup, she is found to have a 5-mm secundum ASD with predominant left-to-right shunt, but does have transient right-to-left shunt. Tests for hypercoagulable disorders were negative. The patient is a Jehovah Witness. According to the AHA guidelines, what would you recommend for management of the ASD?
A. Recommend device closure of the ASD
B. Recommend observation and treatment with Aspirin
C. Recommend observation
D. Recommend observation and treatment with Coumadin
E. Recommend surgical closure
61. What lesion does this patient have (Fig. 4.9)?
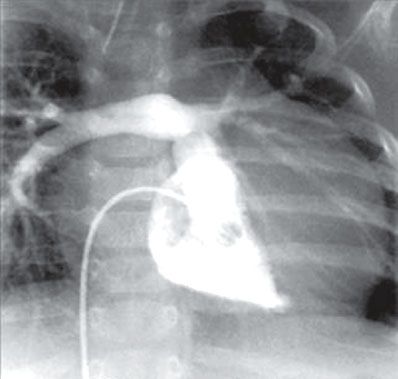
A. Coarctation of the aorta
B. Arterial switch operation
C. Ebstein anomaly
D. Congenitally corrected transposition
E. Senning operation
62. This angiograph in Figure 4.10. demonstrates which of the following?
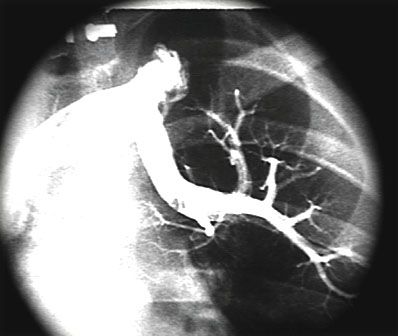
A. Large MAPCA to the left upper lobe
B. Left SVC connecting to the coronary sinus
C. Anomalous connection of the left upper pulmonary vein
D. Pulmonary artery sling
E. Truncus arteriosus with LPA from the descending aorta
63. During a cath in a newborn with suspected congenital heart disease, the saturations in Table 4.4 are obtained.
Table 4.4 Saturations Obtained during a Catheterization in a Newborn

What is the most likely diagnosis?
A. d-TGA, VSD, PS
B. d-TGA, VSD, no PS
C. l-TGA, large ASD
D. d-TGA, PDA, CoA
E. Normal great arteries. PDA + CoA
64. The angiogram in Figure 4.11. demonstrates which of the following?
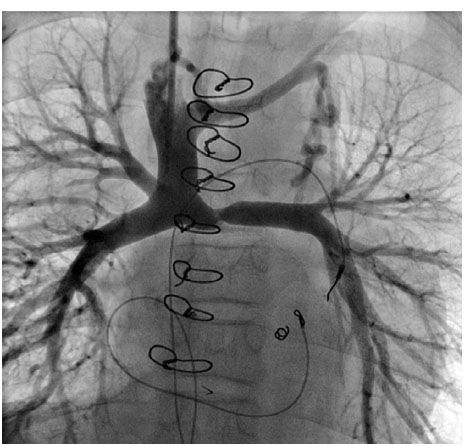
A. BT shunt
B. Sano shunt
C. Bidirectional Glenn
D. Extracardiac Fontan
E. Damus–Kaye–Stansel anastomosis
65. What is the most likely diagnosis in Figure 4.12.?
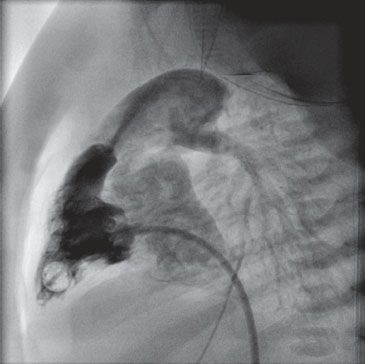
A. VSD
B. ASD
C. PDA
D. Atrioventricular septal defect (AVSD)
E. PS
66. A 3-month-old child with truncus arteriosus is s/p VSD closure and placement of an RV-to-PA conduit. From the angiograph in Figure 4.13., what is the most likely diagnosis?
A. Aneurysm of sinus of Valsalva
B. Aneurysm of RVOT
C. LV false aneurysm
D. Aortico-LV fistula
E. Double-chambered RV
67. What is the diagnosis in Figure 4.14.?
A. Coarctation of the aorta
B. Branch pulmonary artery stenosis
C. MAPCA
D. PDA
E. Double aortic arch
68. In a patient with TOF, which technique will best enhance imaging of the pulmonary artery bifurcation?
A. Cranial angulation of the AP camera with 20 degree LAO
B. Straight lateral, 0 degree position of the lateral camera
C. Extreme caudal position angulation of the AP camera
D. Positioning the angiographic catheter in the RVOT
E. Positioning the angiographic catheter at the RV apex
69. When obtaining vascular access in a neonate. Which statement is correct?
A. Femoral vessels should be entered superior to the inguinal ligament
B. The inguinal crease is the appropriate site for percutaneous access
C. The left femoral vein becomes occluded more easily than the right
D. The right femoral vein is preferred to the left femoral vein
E. Arterial access should be obtained before venous access in all patients
70. Which statement is correct concerning ionic and nonionic contrast media?
A. Ionic agents have a lower osmolarity
B. Nonionic agents cause more physiologic derangement
C. Nonionic agents are less expensive
D. Ionic agents are preferred in pediatric patients
E. Both ionic and nonionic agents yield excellent image quality
71. What does the French size of a catheter refer to?
A. Outer circumference
B. Inner diameter
C. Inner circumference
D. Length of the catheter
E. Outer diameter
72. The following oxygen saturation data were obtained during a baseline hemodynamic study in an intubated 5-year-old patient:
SVC: 72%, RA: 85%, RV: 83%, MPA=83%, RPA=84%, LA=92%, RPV=99%, LPV=83%.
What should you do next?
A. Perform an LV angio to look for a VSD
B. Perform a PA angiogram to evaluate for a pulmonary AV fistula
C. Re-evaluate the endotracheal tube position
D. Continue with the ASD device closure
E. Reverse his sedation
73. Which of the following is considered a flow-directed catheter?
A. Judkins right coronary catheter
B. Gensini catheter
C. Pigtail catheter
D. Berman angiographic catheter
E. Multipurpose catheter
74. Which of the following is the most important factor in determining PVR?
A. Blood vessel radius
B. Blood vessel length
C. Blood viscosity
D. Pulsatility of blood flow
E. Pressure of blood flow
75. Which of the following is the best modality to rule out pulmonary vasoconstriction as an etiology of elevated PVR?
A. Hyperventilation
B. Increased sedation
C. Oxygen administration
D. Fluid bolus
E. General anesthesia
76. The major current advantage in using green dye curves is for assessment of which of the following?
A. PVR
B. LVEDP
C. Decreased cardiac output
D. Small left-to-right shunts
E. SVR
77. Thermodilution measurement of cardiac output is least accurate with which of the following?
A. Small left-to-right shunts
B. Decreased cardiac output
C. Decreased PVR
D. Decreased oxygen saturation
E. Increased pH
78. A 10-year-old patient with Shone syndrome has had three cardiac surgeries. During a catheterization you measure: LVEDP = 24 mm Hg and RVEDP = 22 mm Hg. At this point which test would be most useful to delineate the etiology of the elevated VEDPs?
A. TEE
B. MRI
C. TTE
D. TTE with bubble study
E. Coronary angiography
79. In an unrepaired cyanotic patient who has a hematocrit = 75%, what would be prudent to do before cardiac catheterization?
A. Cancel the procedure: risk of stroke is too high
B. Give the patient a dose of warfarin the evening prior to procedure
C. Admit the patient for IV fluid hydration the evening before procedure
D. No special precautions need to be taken
E. Perform the entire procedure on 100% FiO2
80. An infant has angiography for better delineation of the LVOT. Which views are considered the most optimal?
A. Inject LV with four-chamber and long-axial for vertical and horizontal
B. Inject subaortic area PA and lateral for vertical and horizontal
C. Inject LV with cranial and lateral for vertical and horizontal
D. Inject subaortic area caudal and lateral for vertical and horizontal
E. Inject LV with long-axial and RAO for vertical and horizontal
81. Which of the following is the maximal dose of subcutaneous lidocaine?
A. 1 mg/kg
B. 3 mg/kg
C. 5 mg/kg
D. 10 mg/kg
E. 20 mg/kg
82. Intravenous contrast injection may cause which of the following?
A. Coronary vasoconstriction
B. Decreased myocardial contractility
C. Regional wall motion abnormalities
D. Increased LVEDP secondary to injector pump
E. Increased SVR
83. The maximal total dose of nonionic contrast during cardiac catheterization should not exceed which of the following?
A. 1 cc/kg
B. 3 cc/kg
C. 4 cc/kg
D. 6 cc/kg
E. 10 cc/kg
84. Which of the following is a common drug–side effect combination?
A. Lidocaine—seizures
B. Chlorpromazine—vasoconstriction
C. Nitroglycerine—hypertension
D. Dopamine—bradycardia
E. Ketamine—decreased SVR
85. In a patient without an intracardiac shunt, what is the approximate standard deviation of the PA-SVC difference in oxygen saturation?
A. 1%
B. 3%
C. 7%
D. 10%
E. 15%
86. During a catheterization in a newborn with suspected congenital heart disease, the saturations in Table 4.5 are obtained.
Table 4.5 Saturations Obtained during Catheterization in Newborn with Suspected Congenital Heart Disease

What is the most likely diagnosis?
A. D-TGA, VSD, PS
B. D-TGA, VSD, no PS
C. L-TGA, large ASD
D. D-TGA, PDA, CoA
E. PDA + CoA
87. During a cath the oxygen saturations in Table 4.6 are measured.
Table 4.6 Oxygen Saturations Measured during a Catheterization

What is the Qp:Qs?
A. 1:3
B. 1:2
C. 1:1
D. 2:1
E. 3:1
88. What is an approximate “cut-off” value for the upper limit of normal for a mean PA pressure?
A. 10 mm Hg
B. 20 mm Hg
C. 30 mm Hg
D. 40 mm Hg
E. 50 mm Hg
89. What is an approximate “cut-off” value for the upper limit of normal for an RV-to-PA systolic gradient in the setting of an ASD?
A. 5 mm Hg
B. 10 mm Hg
C. 20 mm Hg
D. 25 mm Hg
E. 35 mm Hg
90. What is an approximate “cut-off” value for the upper limit of normal for RVEDP?
A. 2 mm Hg
B. 5 mm Hg
C. 8 mm Hg
D. 10 mm Hg
E. 12 mm Hg
91. Which of the following lesions would be at highest risk of complication with BAS?
A. HLHS with restrictive atrial septum
B. D-TGA with IVS
C. Tricuspid atresia
D. D-TGA with VSD
E. Total anomalous pulmonary venous connection
92. On which congenital stenotic cardiac valve was the first interventional cath performed?
A. Aortic
B. Tricuspid
C. Mitral
D. Pulmonary
93. Which cardiac valve annulus should NOT be overdistended at balloon valvuloplasty?
A. Aortic
B. Tricuspid
C. Mitral
D. Pulmonary
E. All of above
94. Utilizing a two-balloon technique for pulmonary valvuloplasty, what combined diameter should be used compared to the annulus dimension?
A. 80%
B. 100%
C. 170%
D. 200%
E. None of above
95. Which of the following is LEAST associated with significant branch pulmonary artery stenoses?
A. Edward syndrome
B. Rubella syndrome
C. Williams syndrome
D. Alagille syndrome
Answers
1. ANSWER: D. Oxygen saturation may be derived by using the pH, partial pressure of CO2 (pCO2), and pO2, using the oxygen dissociation curve. The typical O2 dissociation curve is affected by temperature, hydrogen ion, fetal hemoglobin, and levels of 2, 3-DPG. Increasing 2, 3-DPG levels decreases O2 affinity, shifting the dissociation curve to the right, thereby increasing O2 delivery to the tissue. Glucose is not a factor in this calculation.
2. ANSWER: C. Calculation of left-to-right and right-to-left shunts requires understanding the concept of effective pulmonary flow (Qep) and the effective systemic flow (Qes). The Qep is the volume of systemic venous return (i.e., “blue” blood) that flows to the lungs to be oxygenated. Therefore, Qep is calculated by using the oxygen saturation of the “red” blood flowing out of the lungs (pulmonary vein) minus the saturation of the “blue” blood flowing into the lungs (superior vena cava) using the following equation:
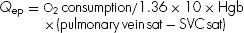
The Qes is the volume of pulmonary venous return (i.e., “red” blood) that flows to the body. For all patients in a steady hemodynamic state:
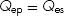
Simply stated, the amount of “blue” blood that flows to the lungs is equal to the amount of “red” blood that flows to the body. If there is no left-to-right shunt, pulmonary artery saturation and SVC saturation are similar, therefore, all the blood flowing to the lungs (Qp) is “blue” blood (Qep). That is:
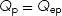
When there is a left-to-right shunt, some oxygenated blood recirculates through the lungs, thus Qp > Qep. The volume of a left-to-right shunt is the difference between the total pulmonary flow (Qp) and the effective pulmonary flow (Qep):
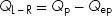
Similarly, when there is a right-to-left shunt, some of the deoxygenated “blue” blood bypasses the lungs and recirculates through the body, thus Qs > Qes. The volume of a right-to-left shunt is the difference between the total systemic flow (Qs) and the effective systemic flow (Qes):
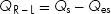
Since Qep = Qes
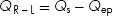
3. ANSWER: B. Simplified mathematically, the ratio of pulmonary flow to systemic flow is Qp/Qs. Since the oxygen consumption is in the numerator of both the Qp and Qs equations, it cancels out, leaving the following equation:
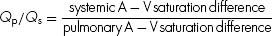
Stated in terms of oxygen saturation samples:

4. ANSWER: C. Accurate leveling of the transducer to reflect the level of the catheter tip is most critical; factors that increase the error in pressure measurement in fluid-filled catheters include air bubbles in the system, smaller caliber catheter, flexible catheters (i.e., elastic tubing may accentuate the pressure, causing fling, or compliant catheters may dampen/smooth the pressure tracings), stopcocks in the system and increased tubing length. A detailed discussion follows:
Eight potential sources of pressure tracing artifact and recommendations for identifying the source of artifact and error are listed below:
1. Loose connections in the system: These usually result in an overdamped tracing. Backup of blood into the transducer tubing is an indication of a loose connection. Air bubbles may be present in the loose connection (see 2 below)
2. Air in the system: This is probably the most common cause of measurement error. Air may be introduced into the system at any of the connections, or dissolved air may come out of the saline used to flush the system. The presence of air in the system lowers the frequency response, producing a damped tracing. Another indication of air in the system is the amplification of high-frequency input, producing overshoot or “fling” in the tracing. The appearance of a small amount of blood with obvious pulsatility in the transducer tubing is an indication that there is air in the system.
3. Inaccurate calibration or baseline drift: Even if the transducers are properly calibrated at the beginning of the procedure, movement of either the patient or the transducers, or electric drift of the baseline, may result in inaccurate pressure recordings. Although small errors in calibration may be inconsequential in arterial recordings, they can have a significant impact in the measurement of venous pressures and PVR.
4. Partial catheter obstruction: This is usually the result of the catheter clotting or kinking. This usually occurs when small or thin-walled catheters are used. If blood is allowed to remain in the catheter lumen for any length of time, deposition of fibrin or platelets will reduce the lumen size, decreasing the frequency response and dampening the pressure.
5. Catheter “fling”: The appearance of fling (a tall, narrow spike) on a pressure recording has many causes. Overshoot can be produced by air in the system. Rapid movement of the catheter tip, which may occur if the tip lies in a turbulent jet, can result in superimposition of high-frequency oscillations on the pressure recording. If the catheter is contacted by a cardiac structure (such as the mitral valve), the superimposed oscillation can alter the waveform dramatically. At times, these conditions are unavoidable.
6. End-hole artifact: When a column of blood stops suddenly against an end-hole catheter, kinetic energy is transformed into pressure energy, and the recorded pressure is falsely elevated. Similarly, when a column of blood is moving away from an end-hole catheter, the pressure recorded will be less than the true intravascular pressure, in proportion to the velocity of flow. This explains why the pressure in a stenotic proximal pulmonary artery has a lower peak systolic pressure than the distal larger vessel.
7. Peripheral pulse wave amplification: Both the peak systolic pressure and the pulse pressure are amplified with increasing distance from the aortic valve. The mean pressure, however, remains the same. This phenomenon is demonstrating by recording the pressure tracing during a pullback around the aortic arch, where the systolic pressure increases from the ascending to descending aorta. While peripheral pulse wave amplification is a physiologic phenomenon and not an artifact, per se, it may lead one to incorrectly interpret pressure data. When comparing the ascending aorta systolic pressure with the femoral or radial artery systolic pressure, failure to take into account pulse wave amplification will result in underestimation of a pressure gradient across an aortic valve or coarctation of the aorta.
8. Catheter entrapment: An end-hole catheter placed in a small or heavily trabeculated ventricle can trap a small volume of fluid, resulting in an exaggerated pressure elevation during systole. To eliminate this error, slightly withdraw the catheter or deflate the balloon (if a balloon-tipped catheter is used). If these maneuvers are not possible, the pressure should be recorded using a side-hole (multipurpose) catheter.
5. ANSWER: B. Mid-chest corresponds to the level of the heart in the chest; this is the most accurate for measurement of intracardiac pressures. The remote transducer is positioned at the side of the catheterization table and positioned at the mid-chest level of the patient; the zero is obtained from this position (half-way between the front of the chest and the back of the thorax). Once attached to the table, this transducer level moves up/down with the patient as the table is moved accordingly. Measurements below the baseline suggest that the transducer is leveled too high.
6. ANSWER: C. The SVC and IVC are extracardiac; typically mean pressures are obtained. RA and LA pressures are measured with calculation of the mean pressure and measurement of the a and v waves (nonphasic); the LV is the only true phasic measurement in this list (systolic and end-diastolic pressure measurements are typically recorded).
7. ANSWER: C. Soft-balloon-tipped catheters are commonly used in pediatric catheterization; wedge catheters have an end-hole, and angiographic catheters have side holes. CO2 is used to inflate balloon as it will be absorbed into the circulation if the balloon ruptures and will not create an air embolus. These catheters are softer, more floppy, and have less torque so are usually quite responsive to direction of blood flow. Since the balloon catheters have two lumens, one for the balloon and one for pressure measurement or contrast injection, the flow rate for contrast injection is not as high as with a single lumen catheter (also meaning this catheter will accommodate only a smaller wire, e.g., 0.025” wire into the 5Fr catheter).
8. ANSWER: E. See question 4 discussion.
9. ANSWER: B. Pressures measured through manometer-tipped catheters (e.g., Millar Instruments Inc., Houston, TX) are the most accurate as there is a direct pressure measurement from the piezoelectric crystal at the catheter or wire tip, and no need for transmission of fluid waves through a fluid-filled catheter to measure the pressure. The errors in pressure measurement from transmission of pressure through a fluid-filled column (also present in Angiographic, end-hole, and balloon-tipped catheters) are avoided. Similar to fluid-filled catheters, there may be artifact due to catheter entrapment, excessive movement in a cardiac chamber, or positioning in a tiny side vessel. These manometer-tipped catheters are less commonly used in pediatric catheterization as they can be challenging to maneuver and do not have a lumen through which to pass a guidewire that may aid in positioning.
10. ANSWER: A. Normal intrathoracic pressure changes during respiration produce a −3 mm Hg to −8 mm Hg negative deflection during inhalation; the most accurate pressure measurements are made at end-exhalation (the passive phase). Deeper breathing will generate more negative intrapleural pressure, and airway obstruction can produce up to 50 mm Hg of negative deflection if very severe.
11. ANSWER: A. The force of the blood flow into the tip of the catheter will increase the pressure measurement by 2 mm Hg to 4 mm Hg; conversely, if a catheter is facing “downstream” in a jet of turbulence, the pressure may be slightly lower, so that when advanced farther beyond this area one may observe the phenomenon of recovery of pressure.
12. ANSWER: A-4, B-1, C-3, D-5, E-2. The “a” wave in a right atrial tracing reflects atrial contraction (systole) and occurs right after the p wave on the ECG. After atrial contraction, the right atrial pressure decreases, this is the “x” descent; during this time a small positive deflection in the decline, the “c” wave, is caused by tricuspid closure (this may be very close to the “a” wave). Following the nadir of the x descent, right atrial pressure then increases due to filling from the systemic venous system and is at a maximum just before the opening of the tricuspid valve. This “v” wave reflects increasing RA pressure during RV systole and is maximal just before opening of the tricuspid valve; it correlates with ventricular systole on the ECG. Once RV pressure is below the level of the RA pressure, the tricuspid valve opens. The “y” descent then reflects the rapid decrease in RA pressure due to RV diastolic filling during opening of the tricuspid valve.
13. ANSWER: B. See questions 12 for a discussion of atrial pressures, which are similar in the left atrium. Usually the “a” wave in the RA is slightly higher than the “v” wave, the converse is true in the LA where the “v” wave is slightly higher than the “a” wave. The “v” wave is due to atrial filling with a closed AV valve during LV contraction; in the setting of mitral regurgitation the v wave will be increased when blood also flows back into the LA through the regurgitant orifice. An increased “a” wave occurs in mitral valve stenosis or LV diastolic dysfunction (a stiff ventricle).
14. ANSWER: D. Mixed venous saturation is the most challenging of these samples if there is a large left-to-right shunt across the ASD due to streaming patterns in the right atrium affecting the right atrial saturation; depending on the location of the ASD this shunt may stream into the IVC or near the SVC. Pulmonary vein saturation is easily measured with direct sampling, with the catheter advanced across the ASD and advanced into the pulmonary vein. PA saturation and systemic saturation are similarly and accurately measured with appropriate catheter position.
15. ANSWER: D. Renal vein oxygen saturation is the highest of these areas mentioned. The IVC receives flow from multiple venous sources, including the renal veins (higher saturation), hepatic veins, and gastrocolic veins (both with lower saturations). The typical IVC saturation is ~5% to 10% higher than the SVC as a result of these venous inflows but may not be consistently observed.
16. ANSWER: A. Coronary artery O2 extraction is the highest in order to supply the myocardial oxygen demands; hence, venous saturation from the coronary sinus (coronary venous drainage) is the lowest of any systemic venous structure entering the right atrium. Coronary sinus return makes up ~5% to 7% of systemic venous return; a typical saturation runs between 25% and 45% and thus can have an impact on mixed venous saturation when there is no shunt lesion.
17. ANSWER: E. Complete mixing of the venous return takes place in the right-sided cardiac chambers; the most distal sample point will provide accurate reflection of mixing when there is no shunt. If the question read “without intracardiac shunt, ” then there is a possibility of shunt at the great vessels, that is, PDA or AP window and the most distal mixed venous sample would be from the right ventricle (in the absence of pulmonary valve regurgitation of significance). Owing to the multiple contributions of systemic veins to the overall flow into the right atrium, even sampling in the RA without an atrial level shunt may result in differences in saturations. In a patient with an intracardiac shunt, the usual location that is used for mixed venous sample is the SVC sample, as this saturation when compared in normal patients without shunt is closest to the pulmonary artery saturation.
18. ANSWER: C. The RLL has the largest vascular distribution of the lobes mentioned, consequently it has the highest blood flow. Regional alveolar and vascular pressures govern perfusion in the lungs, and there is a nonuniform distribution of flow. Traditionally, West proposed a gravitational model where there are three zones of perfusion, stacked vertically, where blood flow is governed by the relationship between alveolar pressure, arterial pressure, and venous pressure, with progressively increased flow present in lower segments of the lung. Effects of gravity may result in alveolar pressure being higher than pressure within the vessels in the upper lobes, resulting in vessel collapse and reduced flow (the zone model again). Other studies have shown this is potentially present in a horizontal stacking fashion (relevance to recumbent and ventilated patients). Detailed studies using other radioactive gases and such have suggested that gravity is not wholly responsible and that this model fails to explain heterogeneity in ventilation/perfusion in the absence of gravitational effects.
19. ANSWER: A. Insure that you are facile with calculation of Qp/Qs using measured oxygen saturations:

The questions on the test may not be straightforward such as the one presented in this table, but imbedded in a longer narrative. The ability to recognize if a shunt is large (Qp/Qs>1.5) thereby impacting operability in patients is needed for these questions. Without the use of calculator in this question, one must be able to quickly calculate the pulmonary to systemic flow ratio.
Pediatric cath labs typically use the SVC saturation as the mixed venous sample in the setting of intracardiac shunts. More precisely, this sample should be obtained from the vessel or chamber just before the level of the shunt. So, the RA sample in the example of a VSD. One must be careful re. the impact of valve regurgitation on shunt calculations as a shunt lesion into a distal chamber may be reflected as a step-up in oxygen saturation in the proximal chamber if the valve in between the two is severely regurgitant. For example, a patient with moderate or severe tricuspid regurgitation may have an elevated RA sat in the setting of a significant shunt at ventricular level. To exclude this factor, many would suggest using the SVC sat as the mixed venous in this clinical situation. In cases where the pulmonary venous saturation is not measured and the systemic arterial blood is not desaturated, most will utilize 97% or the measured systemic arterial saturation also as the pulmonary vein value. In this case we used 95% for the pulmonary venous value.
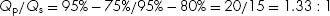
20. ANSWER: D. Again without a calculator be able to determine PVR:
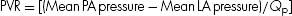
When LA pressure is not directly measured, then pulmonary capillary wedge pressure is the surrogate that should be used. In this case:
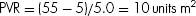
Wood units × 80 = dynes s/cm5
21. ANSWER: E. Using the concepts covered in previous questions,
Qp/Qs = (100 – 85) / (100 – 95) = 15/5 = 3:1 Is this correct?
What did we not account for in this simplified formula?
The patient received supplemental oxygen, hence, the dissolved oxygen needs to be considered. What effect will this have on the shunt volume? (It will reduce the net left-to-right shunt calculation). Using the simplified formula, the calculated shunt = 3.1, but when one considers that shunt will be lower when the dissolved oxygen is factored in, the closest value <3:1 in the given answers is 2.9.
22. ANSWER: D. 0.003 is the constant that is multiplied by the pO2 to determine oxygen content. When the patient is studied in room air, the dissolved oxygen component is not as important. However, when the partial pressure of oxygen is high (pO2 >100 torr), then the dissolved component is important and calculation of Qp/Qs will be reduced.
23. ANSWER: B. While patients typically have higher gradients when brought for BPV, the minimum gradient should be at least 35 mm Hg in a patient who also has RVH. BPV has been used successfully in patients of all ages and is particularly effective in neonates. The preferred position of the exchange wire is the distal left lower pulmonary artery. Since the LPA is usually an extension of the MPA, this position provides a stable straight “rail” to advance a balloon catheter. Positioning in the RPA adds extra turn on the wire, which may enhance losing position if the balloon catheter requires manipulation as it is advanced. The optimal balloon diameter is 1.2 to 1.3 × the annulus dimension. Exceeding 1.4 × the annulus diameter increases the chance of creating significant pulmonary regurgitation. To cause minimal collateral damage to the RVOT and MPA, low pressure balloons (<6 atm) are used for BPV. Low-profile balloons with quick deflation times are particularly useful in neonates with closed ductus. Higher pressure balloons may be used when valves are particularly thick or the annulus dimensions are large.
24. ANSWER: D. BAS involving a quick jerking motion of a septostomy balloon is best performed in a child <1-week old, especially in those with d-TGA and normal size or large left atria. In the first weeks after birth, the atrial septum thickens and the pliability of the septum is reduced. Older children who require enlargement of an atrial septal opening should be managed with balloon dilation, cutting balloon, or blade septostomy techniques. Lesions in which the LA may be small will also pose anatomic challenges. The Rashkind-type of balloon are inflated with hand pressure, jerked briskly but briefly across the septal opening and deflated promptly before the balloon is pulled into the IVC. Simultaneous 2D echocardiographic imaging is recommended for this procedure. A successful tear in the septum is usually signified by an increase in the mobility of the septum primum, laminar color flow across the septum, and a decrease in gradient. In a neonate with d-TGA, systemic arterial oxygen saturation may improve promptly.
25. ANSWER: C. Excellent results for neonatal aortic balloon valvuloplasty have been reported from many centers. The Boston Children’s group recently reviewed their 25-year data (Am J Cardiol, 2011;108:1024–1028). This article also reviews the literature on this topic very nicely and is worth reviewing before taking the board test. Freedom from death or transplant was 87% at 15-year follow-up (answer C). Freedom from repeat valvuloplasty = 61%. Freedom from AVR = 61%. Best results were obtained if post-procedure gradient was <25 mm Hg and in patients with higher pre-procedure LVEF.
26. ANSWER: C. Table 1 in Inglessis & Landzberg. Interventional catheterization in adult congenital heart disease. Circulation. 2007;115:1622–1633 is an excellent review of indications for catheter-based therapy in adults with congenital heart disease. C is the only class 1 indication listed. Asymptomatic adults with mean Doppler or catheter peak-to-peak gradient >40 mm Hg should undergo pulmonary balloon valvuloplasty. Interventions have been recommended for the other answers but they are class 2 indications.
27. ANSWER: D. The posterior/inferior rim (best visualized in the subcostal sagittal transthoracic echocardiography imaging plane) which is close to the IVC/RA junction and the OS of the coronary sinus needs to be adequate (at least 3 mm to 4 mm) in order to stabilize an ASD closure device. The retro-aortic (also known as the anterior/superior) rim frequently is deficient and most of the commercially available devices can straddle the aorta well. A deficient retro-aortic rim may require over sizing of the device by several millimeters in order to engage the aorta effectively and obtain ASD closure. Transcatheter closure devices should not be used in sinus venosus defects (absence of the superior/posterior rim) or primum ASDs (absence of the atrio-ventricular septum) because of interference with important intracardiac structures.
28. ANSWER: E. All the complications have been well reported. However, in the last several decades the frequency of dye allergy has been reduced substantially. The use of nonionic contrast media in the current era has made the incidence of dye allergy 1 in several thousand cases.
29. ANSWER: A. ECG abnormalities after ASD closure device placement have been reported in 5% to 10% of patients. The incidence appears higher in older patients as compared to children. The vast majority of these abnormalities resolve, especially in children, within 24 hours of the procedure.
30. ANSWER: B. During balloon aortic valvuloplasty, the balloon size should not exceed the annulus dimension. The risk of significant post-procedure valve regurgitation may occur if the balloon exceeds 90% of the annulus diameter.
31. ANSWER: D. The general recommendation is that pulmonary balloon valvuloplasty should be performed with a balloon that is 120% of the annulus diameter and should not exceed 140% of the annulus diameter. The risk of significant post-procedure valve regurgitation is increased when the balloon size exceeds 140% of the annulus diameter.
32. ANSWER: A. Pacing the RV rapidly 160 bpm to 200 bpm reduces cardiac output thereby permitting more stable balloon position. This maneuver is commonly performed during aortic balloon valvuloplasty and coarctation of the aorta stent placement. It is also utilized with the newer dynaCT imaging that requires a prolonged slow injection to improve image quality.
33. ANSWER: E. A major component of the Amplatzer device is an alloy (mixture of metals) called nitinol. More than 33% of nitinol is composed of nickel. Detectable levels of nickel have been identified in the bloodstream after successful Amplatzer device placement. The Gore Helex device is composed of a PTFE fabric attached to a nitinol wire.
34. ANSWER: D. Device erosion with the Amplatzer device occurs in ~0.1% of implants. Speculation regarding over sizing and impingement on the wall of the aorta has been debated. However, erosions have been reported with smaller devices (20 mm) also. The Gore Helex device does not appear to be as susceptible to erosion.
35. ANSWER: D. In large patients primary stent placement is recommended for native coarctation versus surgical repair. In small children, balloon angioplasty of native coarctation has a high recurrence rate (perhaps as high as 60% to 65%). This is thought to be due to elastic recoil of the vessel.
36. ANSWER: B. The largest commercially available devices in the US are in the 38 mm to 40 mm range. This defect should accommodate a device easily. Antiplatelet therapy (usually aspirin) is recommended for 6 months after device placement. Device embolization is rare. Most devices can be snared and retrieved without surgical intervention. Transient arrhythmia after device placement is ~10%.
37. ANSWER: C. Use of intravascular for many congenital cardiac lesions has obviated need for surgery in many patients. However, pulmonary venous stenoses remain challenging with poor long-term results from many centers.
38. ANSWER: D. It is doubtful that board questions will appear in 2013 related to the hybrid operation for HLHS, since its use remains center specific and controversial. But one should know the basic concepts of the procedure: Bilateral PA bands to restrict pulmonary blood flow, ductal stent to insure perfusion of the descending aorta and possible atrial septostomy if the PFO is restrictive.
39. ANSWER: B. Intravenous hydration starting the day before the procedure in patients with cyanotic disease and secondary erythrocytosis is reasonable. Patients this age are typically NPO from midnight before the procedure. Many of these patients have bleeding issues. There is no role for prophylactic warfarin. Unless an interventional procedure is planned, there is no need for endocarditis prophylaxis measures. Risk of transient conduction issues during catheter manipulation is not higher in cyanotic patients.
40. ANSWER: A. The patient is acidotic; hence, balloon septostomy should be performed promptly. RV angiography and other diagnostic procedures may be performed later if the patient’s systemic arterial oxygen saturations improve dramatically. Mixing primarily occurs at atrial level in patients with complete TGA, and those with a VSD may still be profoundly cyanotic and acidotic requiring a septostomy.
41. ANSWER: E. See explanation for question 31.
42. ANSWER: D. The step-up occurs at the level of the RV, making one suspect a VSD. However, this is not one of the answers. A large shunt into the PA with associated severe pulmonary valve regurgitation could also explain the RV step-up. The other answers would demonstrate some increase in saturation in the RA. Pulmonary A-V fistula would result in decreased systemic arterial saturation.
43. ANSWER: A. This infant has TOF.
–A. “Cardiac cath should be used in any circumstance in which the anatomy of a child with CHD is inadequately defined by noninvasive means.” ACC/AHA Pediatric Cardiac Catheterization Guidelines, 2011, p. 2609.
–B & E. In an unoperated 6-month-old child, direct calculation of PVR or Qp/Qs is rarely required before surgical intervention.
–C. Only in TOF patients without infundibular stenosis would one consider valvuloplasty. The infundibulum is small in this angiograph.
–D. Many surgeons prefer to leave a PFO or small ASD open when performing TOF repair. The RV, especially in the older TOF patient, is noncompliant thereby elevating RA pressure. The PFO/ASD acts as a “pop-off valve.”
44. ANSWER: C. The data presented demonstrate a markedly elevated LVEDP, not an unexpected finding in a patient with two prior cardiopulmonary bypass exposures. The pulmonary pulse pressure is wide and consistent with severe pulmonary regurgitation. The RA pressure is similar to the RVEDP and not elevated, making severe tricuspid regurgitation unlikely. The final issue to address is this restrictive filling or constrictive physiology (typically observed in patients with cardiac tamponade)? The following features are more suggestive of restriction rather than constriction:
RVEDP > 50 mm Hg, LVEDP – RVEDP > 4 mm Hg, PCWP – mean RAP > 4 mm Hg, RVEDP/RVSP < 0.3
45. ANSWER: D. The chest radiograph demonstrates a heavily calcified pericardium. One would expect this person to have constrictive physiology with elevated LVEDP, mean PCWP, RVEDP, and mean RA pressure. The only answer consistent with elevated intracardiac filling pressures is D. Systemic hypertension is not necessarily a part of constriction; in fact, patients frequently are hypotensive.
46. ANSWER: C. This AP fluoroscopic image demonstrates dextrocardia. The catheter was advanced from a left-sided azygous (hemi-azygos) vein through a small portion of the proximal left-sided SVC into the RA, across tricuspid valve into the RV and then out into the MPA with the tip positioned in the distal LPA. First, note the position of the cardiac silhouette within the chest when trying to determine catheter positions. In addition be careful if images are not true AP or lateral but rather RAO or LAO angled.
47. ANSWER: E. Lateral 0 degree angiograph. The catheter courses from below the diaphragm in a very posterior vessel, in fact, posterior to the aorta. The aorta is typically positioned just anterior to the vertebral bodies. The only vessel listed posterior to this position is the azygous vein. Lack of an intra-hepatic IVC may make interventional catheterization procedures technically challenging.
48. ANSWER: A. A left SVC is demonstrated from the catheter course during this injection of the innominate vein. There is no obvious right SVC. There is cardiac dextroposition and the morphologic RA (receiving the IVC) is likely to the left consistent with situs inversus. The syndrome most commonly associated with situs inversus is Kartagener.
49. ANSWER: B. During interventional catheterization procedures, intravenous heparin 50 units/kg to 100 units/kg is usually administered after vascular access is obtained. Antibiotics are best administered shortly after the patient enters the procedure room. Antiplatelet therapy is instituted after device placement. Subcutaneous low molecular weight heparin does not have a role during the cath.
50. ANSWER: D. The defect is large (23 mm) especially in a small child. The anterior/superior and the posterior rim are small or deficient making device closure unlikely, and this patient likely is best treated with surgical closure.
51. ANSWER: D. The angiograph demonstrates the so-called goose neck deformity in a patient with an AVSD with associated subaortic obstruction due to LVOT elongation. Marfan and Loeys–Dietz are associated with aortic disease. Williams with supra-valve aortic stenosis and branch PA stenosis. Ellis–van Crevald is associated with large (common atrium)-type ASDs.
52. ANSWER: A. In selected patients with pulmonary arterial hypertension who have been unresponsive to medical management, atrial septostomy may be considered to allow preservation of cardiac output in severe pulmonary vascular disease at the expense of increasing systemic desaturation (Evidence: C).
–When blade septostomy is indicated (thickened septum), balloon septostomy should not be performed first as it may stretch rather than tear the septum leading to a temporary interatrial communication. In addition, the stretching of the septum may lead to suboptimal incisions when blade septostomy is performed.
–Vascular injury is a rare complication of atrial septostomy.
–Currently, there are four different catheters that can be used for BAS: Miller catheter (Edwards Lifesciences), Rashkind balloon catheter (USCI-CR Bard), Fogarty (Paul) balloon catheter (Edwards Lifesciences), and the NuMED septostomy catheter (NuMED). The NuMED catheter is the only one with an end hole that enables the operator to advance it over a guidewire and to confirm position by injecting contrast in the left atrium.
–Once the balloon is positioned in the left atrium and the position is confirmed, the balloon is inflated with the appropriate volume of saline/contrast mixture, and a forceful jerk/pull is applied to the catheter to bring the balloon to the junction of the right atrium and inferior vena cava. Care must be exercised as to how vigorously the balloon is pulled into the inferior vena cava. This process is repeated at least once until there is no resistance to passage of the full balloon across the defect. A gradient across the septum may be measured, and if still significant, a BAS may be repeated as above.
53. ANSWER: A. AHA Pediatric Catheterization Guidelines, p. 2627. Ductal stenting may provide acceptable short-term palliation in cyanotic newborns that have another source of pulmonary blood flow, such as patients with critical pulmonary stenosis or pulmonary atresia with intact ventricular septum after RV outflow tract dilation. Such patients often require only short-term additional pulmonary blood flow from the stented duct, and they typically have a straight ductus, which is most amenable to stenting. Patients with a more tortuous ductus (≥2 turns) may also benefit from ductal stenting, but the literature suggests that stenting of such ducts is technically challenging, and the procedural risk–benefit ratio is currently not as favorable. Because restenosis is relatively common, ductal stenting may not be as appropriate in patients who are absolutely dependent on ductal patency for pulmonary blood flow and who therefore require a secure and reliable palliation. Certainly, if such a newborn is deemed to be at too high a risk for a surgical shunt, then ductal stenting may be reasonable. However, very careful follow-up is mandatory, because early and unpredictable restenosis of the stented duct makes such palliation less reliable and potentially hazardous in such a patient.
Class III: Ductal stenting should not be performed in an infant with cyanotic CHD who has obvious proximal pulmonary artery stenosis in the vicinity of the ductal insertion (level of evidence: C).
54. ANSWER: E. AHA Pediatric Catheterization Guidelines, p. 2629. In patients with a large PDA and bidirectional flow due to pulmonary vascular disease, occlusion may be beneficial only if the pulmonary lung bed shows some reactivity to pulmonary vasodilator therapy. These patients should undergo hemodynamic assessment and pulmonary vasoreactivity testing before consideration for ductal occlusion. However, data on this group of patients are scant, and long-term follow-up data are unknown. Should pulmonary vascular disease continue to progress, the ductus will no longer be available to prevent the RV pressures from becoming supersystemic.
Class III: Transcatheter PDA occlusion should not be attempted in a patient with a PDA with severe pulmonary hypertension associated with bidirectional or right-to-left shunting that is unresponsive to pulmonary vasodilator therapy (level of evidence: C)
55. ANSWER: C. Balloon angioplasty of native coarctation may be reasonable in patients beyond 4 to 6 months of age when associated with a transcatheter systolic coarctation gradient >20 mm Hg and suitable anatomy (class IIb; level of evidence: C). Aortic dissection in children with native coarctation is very rare. Risk of aortic dissection is higher in older patients especially in patients with associated vessel calcification. Stent implantation should be the primary intervention once the patient has attained a size that a stent that can be re-dilated to normal adult size can be implanted.
56. ANSWER: C. C is a class I guideline for balloon angioplasty. A is not recommended in patients under 4 months of age. There is only class IIb evidence to perform angioplasty in patients with Turner syndrome, hence not the best answer choice. No intervention if not hypertensive and transcatheter gradient <20 mm Hg. In neonates: Class IIa recommendation if patient is unstable, but no indication that this neonate is unstable (see Table 4.7).
Table 4.7 Recommendations for Transcatheter Balloon Angioplasty of Coarctation/Recoarctation of the Aorta

57, 58. ANSWERS: 57-C; 58-B
Coarctation:
Incidence ~6% to 8% of CHD.
Most common associated cardiac anomaly = bicuspid aortic valve (~30% to 40%).
Most important noncardiac anomaly = intracerebral aneurysm ~10%.
Native coarctation treatment gold-standard is still surgical repair (debatable unlikely to test this directly). Some centers using balloon angioplasty with stent placement as primary intervention.
Small incidence of aneurysm formation at angioplasty site in native coarctation.
Recoarctation:
Treatment standard for most young patients = balloon angioplasty.
Long-term results with stent placement not yet known but recoarctation appears less.
Increased incidence of re-intervention need as patient grows.
Incidence of aneurysm less than for balloon angioplasty alone.
In this patient, a head CT or MRI should be completed to rule out cerebral aneurysm. As for the coarctation, if the cath systolic gradient is >20 mm Hg, balloon angioplasty +/− stent placement should be considered.
–Must be able to expand stent to 2 cm.
–Of note, patient would qualify if <20 mm Hg gradient and systemic hypertension without other reason for HTN (Class IIa).
59. ANSWER: C. Patients with PA-IVS have increased risk of coronary circulation abnormalities such as sinusoids, fistulae, coronary stenosis, or atresia.
• RV to coronary artery fistulae occur in 31% to 68% of PA-IVS. RV-dependent coronary circulation (significant portion of LV is supplied by fistulae fed by hypertensive RV) 3% to 34%.
• Decompression of RV in the setting of significant RV-dependent myocardial circulation will result in irreversible myocardial ischemia and is usually fatal.
• Pulmonary valvuloplasty with RV-dependent coronary circulation is a class III recommendation.
• See Table 4.8.
Table 4.8 Recommendations for Pulmonary Valvuloplasty

60. ANSWER: A. See Table 4.9. Device closure is a class IIa recommendation with level of evidence B.
Table 4.9 Recommendations for Transcatheter Device Closure of Secundum ASD

61. ANSWER: D. The angiogram demonstrates a morphologic left ventricle that is connected to the pulmonary arteries. There is atrioventricular and ventricular great artery discordance or congenitally corrected TGA. Many of these patients have abnormal tricuspid valves with “Ebstein” features—but that is not depicted in this angiogram.
62. ANSWER: C. The left upper pulmonary vein was injected through a catheter that courses through the innominate vein. There is anomalous connection of the left upper pulmonary vein to the innominate vein via a vertical vein.
63. ANSWER: E. Ascending aortic saturation is normal. The descending aorta is being perfused primarily by the ductus. Severe coarctation or interrupted aortic is likely.
64. ANSWER: C. A right-sided Glenn connection is demonstrated with PA branch stenosis just to the midline of the anastomosis. You should be able to identify the typical angiographs of the shunts/connections listed in this question.
65. ANSWER: E. Lateral projection RV angiograph demonstrates a narrow jet across the pulmonary valve. You should be able to identify typical angiographs of the lesions listed in this question.
66. ANSWER: B. Lateral projection RV angiograph. Severe aneurysmal dilation of the RVOT is demonstrated, a known complication of RV-PA conduit placement.
67. ANSWER: D. Lateral projection angiograph with injection in the upper descending aorta demonstrating a PDA filling the MPA. Hint for all angiograph questions determines catheter course and where dye is being injected before assessing the lesion.
68. ANSWER: C. Extreme caudal angulation will assist in imaging the PA bifurcation in patients with tetralogy. Cranial angulation of the AP camera is useful for patients with normal anatomy but not always work in tetralogy. The straight (0 degree) lateral projection images the LPA well but overlaps the RPA at the bifurcation. The catheter should ideally be positioned just beyond the bifurcation, so that it fills the area of interest. Injection in the RV will not always adequately image the PA bifurcation especially in large patients.
69. ANSWER: D. The right femoral vein provides straight access to the RA through the IVC. This is particularly important when considering interventional procedures. Access should be obtained 1 cm below the inguinal ligament in a neonate in order to have proper hemostasis after sheath removal.
70. ANSWER: E. Nonionic contrast agents are recommended for use in pediatric patients. Advantages of nonionic contrast include low osmolarity, less hemodynamic derangement, fluid shifts, reflex tachycardia, less elevation of PA and LA pressures, and less exacerbation of pulmonary hypertension. Nonionic agents are more expensive. However, both ionic and nonionic provide excellent image quality.
71. ANSWER: A. French size = outer circumference. A 5 French catheter has an outer circumference of 5 mm, corresponding to an outer diameter of 1.67 mm. The inner lumen diameter will vary regardless of French size. Lumen diameter is usually designated by the wire that will be accommodated, for example, “0.035 cm wire.”
72. ANSWER: C. The left pulmonary venous saturations are unexpectedly low. The right pulmonary veins are fully saturated. If the ET tube is in the right mainstem bronchus, the left lung may be ineffectively ventilated and oxygenated.
73. ANSWER: D. The only catheter with a balloon (thereby flow-directed).
74. ANSWER: A. Resistance = length × viscosity / radius4. Radius is to the 4th power.
75. ANSWER: C. “Oxygen administration often is the only agent needed to determine whether elevated PVR is caused by pulmonary vasoconstriction.” Science and Practice. 2nd ed. p. 980.
76. ANSWER: D. Small left-to-right shunts (<10% to 15%) are difficult to measure by oximetry, but green dye curves can accurately measure them using double sampling techniques.
77. ANSWER: B. “Thermodilution cardiac outputs are less reliable whenever there is very low CO.” Science and Practice. 2nd ed. p. 988.
78. ANSWER: B. “Congestive heart failure, restrictive cardiomyopathy, and pericardial constriction can cause all diastolic pressures to be elevated.” Science and Practice. 2nd ed. p. 966. The difference between the LVEDP and RVEDP is <4 mm Hg, suggesting constriction. It is important to evaluate the pericardium in patients with suspected constriction. This is especially important in postoperative patients. MRI or CT would be reasonable at this point in the evaluation.
79. ANSWER: C. Hydration for patients with elevated Hct. is prudent since renal insufficiency may be exacerbated by prolonged NPO status and angiography.
80. ANSWER: E. Science and Practice. 2nd ed. p. 1008.
81. ANSWER: C. “Convulsions may occur if the total dose of lidocaine exceeds 5 mg/kg.” Science and Practice of Pediatric Cardiology. 2nd ed. p. 998.
82. ANSWER: B. “When contrast material reaches the coronary circulation, there is coronary vasodilation and chelation of calcium from the blood and probably the intracellular fluid. There is a drop in coronary hematocrit due to an increase in intravascular water from the tissues, this results in decreased myocardial contractility.” The Science and Practice. 2nd ed. p. 1003.
83. ANSWER: D. “Allowing up to 6 ml/kg of nonionic contrast material to be given over a period of 2 to 3 hours.” Science and Practice. 2nd ed. p. 1004.
84. ANSWER: A. “Convulsions may occur if the total dose of lidocaine exceeds 5 mg/kg.” The Science and Practice. 2nd ed. p. 998.
85. ANSWER: B. “The standard deviation of the pulmonary artery – superior vena cava difference was 2.9%…” Moss and Adams. 7th ed., vol. 1. p. 217.
86. ANSWER: D. Descending aortic saturation is higher than ascending; it suggests coarctation with PDA. But also TGA because PA sat is high.
87. ANSWER: E. 98–67/100–90 = 3:1. See previous shunt calculation explanations.
88. ANSWER: B. “The normal pulmonary artery systolic pressure is equal to the right ventricular systolic pressure (<30mmHg), and the mean pressure is < 20mmHg.” Moss and Adams. 7th ed., vol. 1. p. 215.
89. ANSWER: B. “Gradients < 10mmHg may be seen with structurally normal pulmonary valves and increased pulmonary blood flow, as with a large atrial septal defect.” Moss and Adams. 7th ed., vol. 1. p. 215.
90. ANSWER: C. “Normally, the peak systolic pressure is < 30mm Hg, and the end diastolic pressure is < 8 mmHg.” Moss and Adams. 7th ed. vol. 1. p. 215.
91. ANSWER: A. BAS is performed to improve atrial mixing (simple d-TGA or d-TGA with VSD) or to treat atrial septal defect restriction in the setting of significant obstruction or stenosis of an atrioventricular valve (HLHS being the most common). It is rarely needed in right heart obstructive lesions, that is, tricuspid atresia (one must be aware that performing a balloon septostomy in a redundant atrial septum, such as in tricuspid atresia, is challenging and one may disrupt the RA/IVC junction with aggressive techniques). It is not performed in the patient with TAPVR. This is a lesion that will be surgically corrected at the time of diagnosis and the atrial septum is not typically the location of “obstruction” of the venous return pathway.
The procedure is usually straightforward in patients with d-TGA where the flap of the fossa ovalis is thin and easy to tear with the balloon catheter. BAS can be performed up to a few weeks of age; after this time the atrial septum may be too thick to balloon successfully in any lesion without significant risk of disruption of normal tissue. A blade septostomy will then be needed. The exception to this is the neonate with HLHS and restrictive atrial septum, where the tissue may be very thick, with minimal or no opening at the time of delivery. This procedure is usually performed emergently and immediately after delivery. It may require various techniques, including transseptal or radiofrequency access to the left atrium, the use of dilation balloon (cutting balloon), and stent placement for complete relief of obstruction. A simple balloon septostomy in HLHS with thick, restrictive atrial septum may avulse the pulmonary veins (i.e., the tissue with the least resistance will give way) during the BAS.
92. ANSWER: D. Balloon angioplasty for pulmonary valve stenosis was first reported by Kan and is now considered standard of care for treatment of pulmonary stenosis.
93. ANSWER: A. Accurate measurement of the annulus is key for any valve intervention. Typically a balloon diameter of 0.9–1.0 is used for aortic valve dilation. It is critical to not oversize a balloon when dilating the aortic valve, as aortic insufficiency may result and it can be catastrophic in the setting of compromised left ventricular function. For mitral valve dilation the Inoue balloon is typically used and the formula to calculate the size of the balloon is based on the patient’s height [balloon size = (height in cm/10) +10]. If the double balloon technique is used the combined diameter of the 2 balloons is usually 130% of the largest measured annulus diameter. Dilation of congenital tricuspid stenosis is extremely uncommon as the lesion itself is rare and usually not an isolated issue. One would use a combined double balloon diameter of ~150% of the measured tricuspid annulus if this procedure is necessary. Pulmonary valve dilation was discussed in other questions.
94. ANSWER: C. Double balloon technique may allow better hemodynamic stability during inflation as the minor gap between two balloons may allow some antegrade flow even at full inflation. As recommended by Mullins, the combined diameter for two balloons would be 1.6–1.8 times the diameter of the pulmonary annulus. Single balloon technique for pulmonary valvuloplasty typically involves using a balloon 20% to 40% larger than the annulus.
95. ANSWER: A. Typical syndromes in which the patient may have peripheral pulmonary branch stenosis include Congenital Rubella, Williams, and Alagille. Edward syndrome (trisomy 18) is typically associated with VSD and valve abnormalities (polyvalvular).
SUGGESTED READINGS
AHA Pediatric Catheterization Guidelines: Feltes TF, Bacha E, Beekman RH III, et al. Indications for cardiac catheterization and intervention in pediatric cardiac disease: a scientific statement from the American Heart Association. Circulation. 2011; 2613–2614.
Allen HD, Driscoll DJ, Shaddy RE, et al. Moss and Adams’ Heart Disease in Infants, Children and Adolescents. 7th ed. Lippincott Williams and Wilkins; 2008.
Brown LK, et al. Anatomic landmarks for use when measuring intracardiac pressures with fluid-filled catheters. Am J Cardiol. 2000;86(1):121–124.
Feltes TF, Bacha E, Beekman RH III, et al. Indications for cardiac catheterization and intervention in pediatric cardiac disease: a scientific statement from the American Heart Association (Guidelines). Circulation. 2011;2621–2623.
Galvin I, Drummond GB, Nirmalan M.Distribution of blood flow and ventilation in the lung: gravity is not the only factor. Br J Anaesth.2007;98(4):420–428.
Garson A, Bricker TJ, Fisher DJ, et al. (eds). The Science and Practice of Pediatric Cardiology. 2nd ed. Williams and Wilkins; 1998.
Glenny RW. Determinants of regional ventilation and blood flow to the lung. Int Care Med.2009;35(11):1833–1842.
Inglessis I, Landzberg MJ. Interventional catheterization in adult congenital heart disease. Circulation. 2007;115:1622–1633.
Kan JS, White RI Jr, Mitchell SE, Gardner TJ. Percutaneous balloon valvuloplasty: a new method for treating pulmonary-valve stenosis. N Engl J Med. 1982;307:540–542.
Lock JE, Keane JF, Perry SB. Diagnostic and Interventional Catheterization in Congenital Heart Disease. 2nd ed. Kluwer Academic Publishers, 2000.
Mullins CE (ed.) Cardiac Catheterization in Congenital Heart Disease. Blackwell Futura, 2006.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


