Chapter 21 Arrhythmias
Disorders of cardiac rhythm are common in the intensive care unit (ICU). Life-threatening arrhythmias such as ventricular tachycardia and complete heart block require immediate intervention. Even arrhythmias such as atrial fibrillation can cause marked hemodynamic instability in cardiac surgery patients. Arrhythmias are commonly a manifestation of underlying cardiac pathology; ventricular fibrillation may be due to acute myocardial ischemia, whereas atrial fibrillation may reflect raised atrial pressure due to ventricular dysfunction. Thus, it is essential that treatment be directed to the underlying cause as well as to the suppression of the arrhythmia.
In this chapter the pathophysiology, diagnosis, and treatment of cardiac arrhythmias, including the use of temporary pacing for the treatment of bradyarrhythmias, are reviewed. The physiology of cardiac conduction and the pharmacology of antiarrhythmic agents are discussed in Chapters 1 and Chapter 3, respectively. The interpretation of an electrocardiogram (ECG) is reviewed in Chapter 8.
PATHOPHYSIOLOGY OF TACHYARRHYTHMIAS
Enhanced and Abnormal Automaticity
Cells of the sinoatrial (SA) node and around the atrioventricular (AV) node normally undergo spontaneous depolarization during phase 4 of the action potential. Any factor that increases the slope of phase 4 (such as β1-adrenoreceptor stimulation) causes the threshold potential to be reached earlier, increasing the rate of action-potential generation (see Fig. 1-2). This is called normal automaticity. If the firing rate of a subsidiary pacemaker is abnormally increased such that sinus node dominance is usurped, it is termed enhanced automaticity, and accelerated junctional rhythm is an example of this. When cells that are normally quiescent become partially depolarized, and resting membrane potential rises to −60 or −50 mV, pathologic pacemaker activity may occur. This is called abnormal automaticity, and it is the basis of some atrial tachycardias.
Reentry
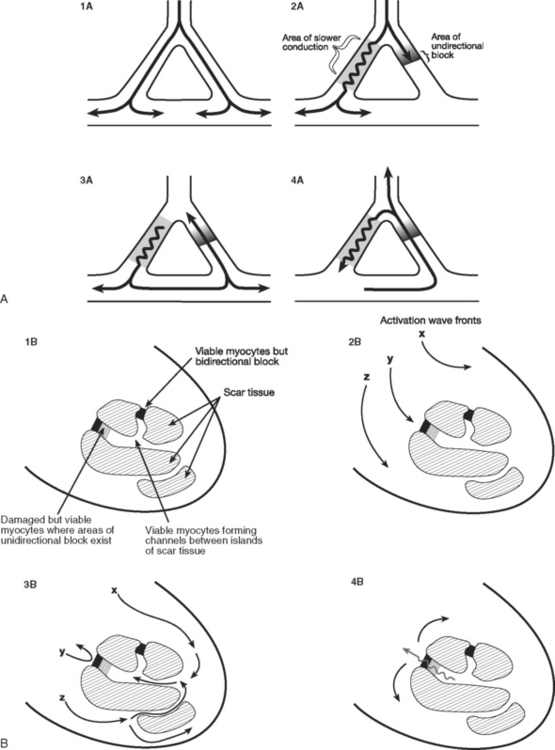
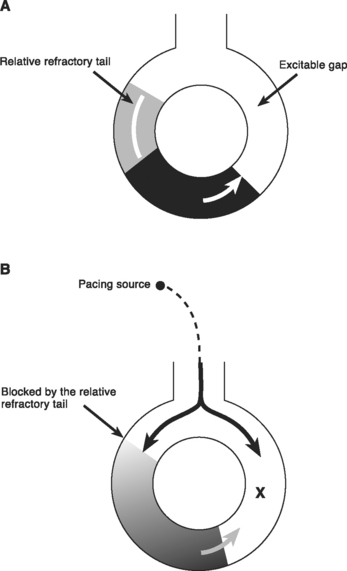
Reentry is the basis of most arrhythmias encountered clinically. Reentry is continuous circulating electricity in which an impulse reenters and repetitively excites a region of the heart. Three requirements are necessary for reentry to occur: (1) an abnormal electrical circuit; (2) slow conduction; and (3) unidirectional block (Fig. 21-1). These conditions may arise because of a fixed anatomic structure such as that which occurs in the Wolff-Parkinson-White syndrome, but more commonly, the conditions for reentry arise due to abnormal physiologic states. For instance, dynamic circuits with unidirectional block may develop due to variability in recovery times (dispersion of refractoriness) among various regions of myocardium. Slow conduction may occur because of partial membrane depolarization or loss of gap junctions.
Proarrhythmic Factors
Hypokalemia
Hypokalemia decreases the resting membrane potential (i.e., it becomes less negative), which would be expected to increase the likelihood of spontaneous depolarization. However, a more important effect is that hypokalemia decreases the permeability of the membrane to potassium, which prolongs repolarization and increases the dispersion of recovery times, thereby increasing the likelihood that a reentry circuit will occur. Hypokalemia is associated with a wide range of arrhythmias, including atrial and ventricular extrasystoles, atrial fibrillation, and torsades de pointes ventricular tachycardia. The ECG signs of hypokalemia are described in Chapter 32.
Myocardial Infarction
The mechanism by which ventricular tachycardia is propagated late after myocardial infarction is quite different from the mechanism responsible for its generation immediately after the onset of ischemia. Within the first 10 minutes of acute ischemia, ventricular tachycardia and fibrillation may occur due to reentry allowed by slowed conduction and unidirectional block in ischemic tissue. In the days that follow myocardial infarction, infarcted myocardium is gradually replaced by scar tissue. Filaments of surviving myocardium encased in this scar tissue show slowed conduction, partly as a consequence of altered membrane potential and partly due to altered architecture and increased intracellular resistance. If unidirectional entrance block occurs at one site on the scar border, with subsequent delayed emergence of a different wave front, ventricular tachycardia may ensue when adjacent, fully repolarized normal myocardium is then immediately reactivated (or depolarized; see Fig. 21-1). Removal of this scar-related circuit is the basis of the “endocardial peel” operation.
NARROW-COMPLEX TACHYCARDIAS
Narrow-complex tachycardia may be defined as a heart rate greater than 100 per minute and a QRS duration less than 0.12 seconds (3 small squares on the ECG). Narrow-complex tachycardias activate the ventricles in a normal manner unless bundle branch block is present (see later material).
Approach to the Patient With Narrow-Complex Tachycardia
A careful review of the 12-lead ECG provides important clues to the diagnosis:
Atrial Electrocardiogram
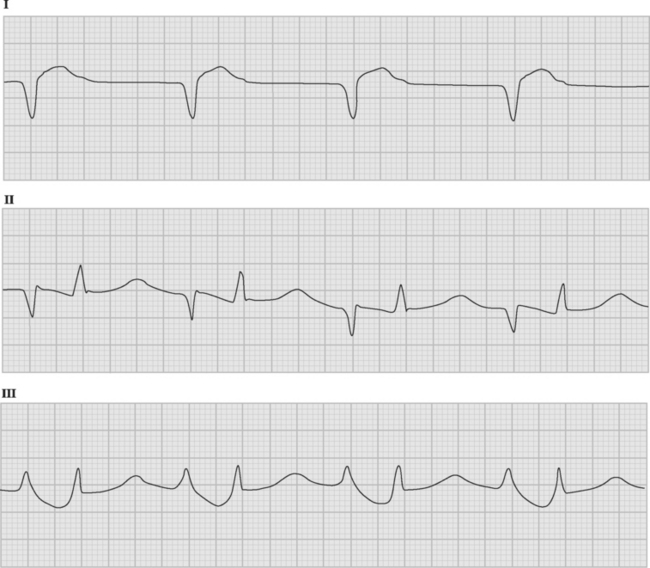
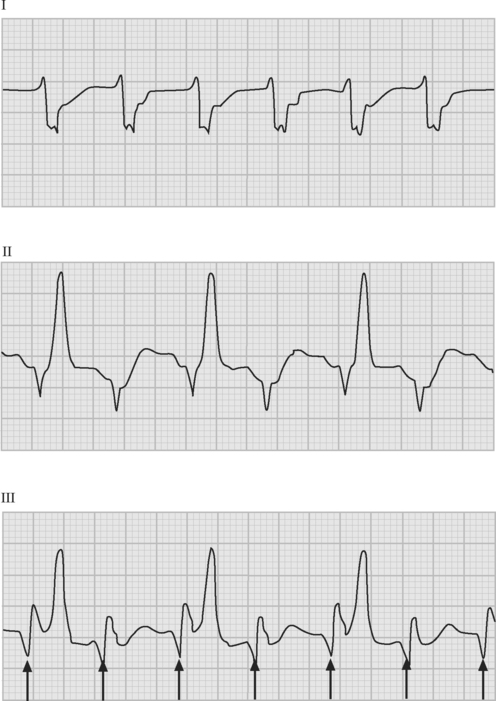
If temporary epicardial pacing wires are in place, an atrial ECG can be performed to clarify the relationship between the P wave and the QRS. One method involves connecting the left and right arm ECG cables to the atrial pacing wires. The left and right lower limb cables are connected normally, and leads I, II, and III are recorded at 50 mm/sec (Fig. 21-2). P waves appear as the dominant complexes in lead I (the bipolar atrial lead), whereas the QRS is usually dominant in leads II and III (the unipolar atrial leads). Alternatively, a single upper limb lead can be connected to an atrial pacing wire and a unipolar atrial lead examined on the monitor: if the left arm cable is used, lead I or III is examined; if the right arm cable is used, lead I or II may be examined.
Atrial Fibrillation
Atrial fibrillation following cardiac surgery is very common, with most series reporting an incidence of between 30% and 60%.1,2 The peak incidence is on the second postoperative day. Postoperative atrial fibrillation is not benign; it prolongs ICU and hospital stay, increases costs, and is associated with an increased incidence of stroke.2
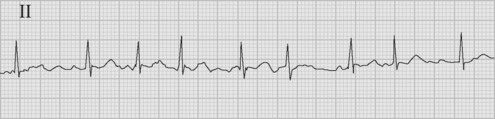
The arrhythmia is characterized by the abrupt onset of an irregularly irregular narrow-complex tachycardia (Fig. 21-3), often with varying R wave amplitude. Heart rate varies widely, depending on adrenergic tone, but is typically between 120 and 160 beats per minute. In cardiac surgery patients, the onset of atrial fibrillation may cause marked hypotension.
Pathophysiology
Atrial fibrillation was regarded for a long time as a classic example of random reentry due to multiple wandering wavelets.1 However, it is now recognized that in many patients the atria are only passively involved and that the initiating trigger is rapid focal discharges from cells located in the muscle sleeves of the pulmonary veins.3
Sustained atrial fibrillation leads to mechanical and electrical remodeling of the atria, with loss of contractility (atrial stunning) and shortening of refractory periods. Atrial stunning persists for several weeks after reestablishment of sinus rhythm, increasing the risk for atrial thrombus formation, especially in the left atrial appendage. The shortening of atrial refractory periods results in reinforcement of the arrhythmia the longer it remains untreated.4 Furthermore, patients are very vulnerable to reinduction of atrial fibrillation immediately after reversion to sinus rhythm. For these reasons, after successful cardioversion of atrial fibrillation of more than 48 hours’ duration, anticoagulation and antiarrhythmic therapy should continue for at least 30 days.5
Risk Factors
The principal risk factor for atrial fibrillation after cardiac surgery is increased age.2 Previous atrial fibrillation, mitral and aortic surgery, atrial enlargement, and withdrawal of β blockers are also important risk factors. Other factors associated with the development of atrial fibrillation include the requirement for inotropes or vasopressors, hypomagnesemia, prolonged cardiopulmonary bypass, and impaired ventricular function. Acute atrial stretch due to impaired ventricular function or tamponade may precipitate atrial fibrillation. Thus, atrial fibrillation may be both a cause and a consequence of hemodynamic instability.
Prophylaxis
β Blockers (metoprolol, carvedilol, and sotalol) and amiodarone reduce the incidence of atrial fibrillation by as much as 50%.6–9 The combination of amiodarone and a β blocker appears to be more effective than either agent alone.10 Other drugs such as magnesium and diltiazem may also be useful for prophylaxis but data are inconclusive.9 Standard (right atrial) pacing may have a small protective effect against atrial fibrillation, but the benefit is greater if simultaneous left and right atrial pacing is used.11,12 Digoxin is not indicated for prophylaxis of postoperative atrial fibrillation.7,9
Prophylaxis against atrial fibrillation should include avoiding β-blocker withdrawal in patients chronically treated with them. For patients at increased risk for atrial fibrillation, routine prophylaxis with a β blocker or amiodarone is appropriate.9 Prophylactic biatrial pacing should also be considered in high-risk patients.12
The preoperative administration of 600 mg per day of amiodarone for 1 week prior to surgery has been shown to be effective8 but may not be feasible. A practical alternative is intraoperative dosing of 5 mg/kg intravenously followed by oral treatment of 400 mg three times daily for 2 days, 200 mg twice daily for 2 days, and then 200 mg daily until hospital discharge. Amiodarone should be withheld in the presence of sick sinus syndrome, bradycardia (heart rate <50 beats per minute), or marked first-degree AV block (PR interval >0.30 seconds). In patients with paroxysmal atrial fibrillation who are chronically treated with amiodarone, low-dose oral sotalol (40 to 80 mg twice daily) may be commenced postoperatively, with careful monitoring for excessive bradycardia or QT prolongation. When atrial fibrillation is associated with planned mitral valve surgery, a surgical maze procedure should be considered (see Chapter 10).
Treatment
Atrial fibrillation that is associated with hemodynamic compromise should be treated aggressively. The success rate for primary cardioversion is low—less than 20% in one study.13 The success rate of electrical cardioversion may be increased by the use of biphasic energy (with an energy level of 200 joules) and the prior correction of hypokalemia and hypomagnesemia. If atrial pressures are elevated, an echocardiogram should be considered to rule out ventricular dysfunction and pericardial tamponade. If cardioversion is unsuccessful, it may be repeated following intravenous amiodarone (a bolus dose of 5 mg/kg over 30 minutes or an infusion of 2 mg/min for 4 hours). Amiodarone provides acute control of heart rate through its β-blocker actions and may itself result in pharmacologic cardioversion. Following successful cardioversion, an infusion of amiodarone (1 mg/min) should be administered until the patient is able to take oral amiodarone or a β blocker. Atrial pacing (AAI mode at 80 to 90 beats per minute for 24 to 48 hours) should be instituted to minimize the chance of recurrence. Intravenous sotalol or ibutilide may be used as an alternative in patients with good left ventricular function.14 If, despite these measures, the patient remains in rapid atrial fibrillation, pharmacologic control of heart rate may be achieved by an intravenous β blocker or diltiazem.15 In patients with unstable hemodynamics, intravenous digoxin may be tried, but it has limited efficacy in the setting of heightened adrenergic tone.15
Pharmacologic treatment for atrial fibrillation should be continued for 4 to 6 weeks following surgery, even if the patient reverts to sinus rhythm.15 If atrial fibrillation persists at 6 weeks following surgery, a choice should be made between repeat cardioversion and permanent rate control and anticoagulation. The latter strategy is favored by the Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) study, which did not show a better outcome for older patients with attempted maintenance of sinus rhythm by antiarrhythmic drugs.16
Cardioversion Guided by Transesophageal Echocardiography
Cardioversion should not be performed without the guidance of transesophageal echocardiography in patients who have experienced atrial fibrillation for more than 48 hours and are not undergoing therapeutic anticoagulation. This is so because of the risk for systemic embolization of intracardiac thrombi.17 If left atrial thrombus is present on transesophageal echocardiography (TEE) examination, the patient should be given anticoagulants with heparin and warfarin, and cardioversion should be performed 4 weeks later if repeat (TEE) shows resolution of the thrombus. If thrombus is not present, cardioversion may proceed immediately, after which the patient should undergo anticoagulation with warfarin for 4 to 6 weeks (see subsequent material).
Anticoagulation for Atrial Fibrillation
If there are no contraindications, patients with sustained or paroxysmal atrial fibrillation for more than 24 to 48 hours after cardiac surgery should be given an anticoagulant with warfarin, aiming for an International Normalised Ratio (INR, see Chapter 30) between 2 and 3. Warfarin should be continued for 4 to 6 weeks following surgery, at which time, if the patient is in sinus rhythm, it may be stopped.5 However, if there are factors predictive of recurrence, such as a history of paroxysmal atrial fibrillation, a left atrial diameter greater than 5 cm, or mitral valve surgery, warfarin should be continued.
Patients at increased risk for thromboembolic complications, notably those with documented atrial thrombus, a history of systemic embolization, or a prosthetic mitral or aortic valve, should also be given anticoagulants with unfractionated heparin, which should be continued until the patient has a therapeutic INR.5 Compared with warfarin, aspirin is less effective in reducing stroke frequency in patients with atrial fibrillation,18,19 and it is therefore not usually an acceptable alternative.
Nonpharmacologic Treatment of Atrial Fibrillation
A number of catheter-based techniques involving isolation of the triggering foci around the pulmonary veins and ablation of fractionated atrial potentials have been developed for the treatment of chronic atrial fibrillation.20,21 In older patients, radiofrequency ablation of the bundle of His with permanent pacemaker implantation provides good symptom relief, although the need for anticoagulation remains. These techniques have a minimal role in the management of postoperative atrial fibrillation.
Atrial Flutter
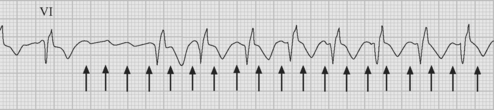
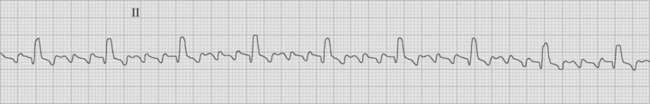
Atrial flutter is usually caused by a reentry circuit around the tricuspid valve with an area of slow conduction near the orifice of the coronary sinus, which results in abnormal flutter waves on the ECG that have a characteristic sawtooth pattern at a rate of between 250 and 330 per minute. Because of the long refractory period of the AV node, usually only a proportion of the flutter waves are conducted to the ventricles; typically in a ratio of 2:1 (Fig. 21-4), resulting in a heart rate of about 150. Higher ratios of AV block (e.g., 3:1 or 4:1) resulting in slower heart rates are also seen (Fig. 21-5). Postoperative atrial flutter typically has 2:1 conduction. If antiarrhythmic drugs have been given as part of routine prophylaxis, the ventricular rate with 2:1 block may be less (125 to 150 beats per minute). Atrial flutter with 2:1 AV block can be difficult to distinguish from other, regular narrow-complex tachycardias because one of the flutter waves may be concealed within the QRS complex; the rhythm may be mistaken for sinus tachycardia with first-degree AV block. Negative flutter waves on the inferior ECG leads and a fixed heart rate of 140 to 150 per minute usually confirm the diagnosis, but if doubt persists, an atrial ECG should be performed if possible (Fig. 21-6). Alternatively, adenosine (6 or 12 mg) as a rapid intravenous bolus will cause temporary AV block, exposing the characteristic flutter waves.
Treatment
The risks for thromboembolism with atrial flutter are no different from those with atrial fibrillation, and the same indications for anticoagulation and cardioversion guided by TEE apply. Typical atrial flutter can be permanently prevented at the time of cardiac surgery by creating a surgical or radiofrequency scar line between the tricuspid annulus and the orifice of the inferior vena cava, extending from endocardium to epicardium (see Fig. 10-8).22
Technique of Overdrive Pacing for Atrial Flutter
Within a reentry circuit, at any point in time a portion of the circuit is not being activated. This is the distance between the advancing head of the circulating wave front and the tail of depolarized cells; it is called the excitable gap. Pacing impulses can invade the circuit via this gap and, by colliding with the arrhythmia wave front, eliminate the arrhythmia (Fig. 21-7). This technique is known as overdrive pacing and is useful for certain reentry arrhythmias, including ventricular tachycardia and AV nodal reentry tachycardia, but it is particularly useful for atrial flutter.
Accelerated Junctional Rhythm
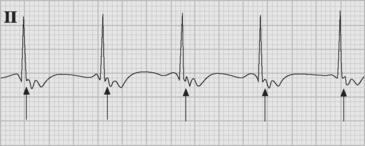
Accelerated junctional rhythm (nodal tachycardia) is a regular narrow-complex tachycardia in which no P wave can be seen preceding the QRS complex. The tachycardia typically develops gradually (warm up), slowly increasing up to a heart rate of 110 to 150 beats per minute. Retrograde-conducted P waves that follow the QRS are visible on an atrial ECG and may be seen on a standard rhythm strip (Fig. 21-8). Depending on the ventriculoatrial interval, regular cannon A waves may be visible on the central venous pressure (CVP) trace (see Chapter 8). Occasionally, the atrial ECG demonstrates AV dissociation, in which case irregular cannon waves are seen instead.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree