The aim of the present study was to investigate the full range of aortic root diameters and stiffness in a group of subjects without known cardiovascular risk factors and/or overt cardiovascular disease. Four hundred and twenty-two healthy subjects (mean age 44.35 ± 16.91 years, range 16 to 90, 284 men [67%]) underwent comprehensive transthoracic echocardiography. The leading edge method was used for the end-diastolic aortic root diameters measured at 4 locations (1) the aortic annulus, (2) the sinuses of Valsalva, (3) the sinotubular junction, and (4) the maximum diameter of the proximal ascending aorta. Aortic wall stiffness was assessed using 2-dimensional guided M-mode evaluation of systolic and diastolic aortic diameter, 3 cm above the aortic valve. The absolute aortic root diameters increased with age in both genders. Aortic measurements were significantly greater in men than in women at all levels, whereas body surface area–indexed values were similar in men and women, except for the ascending aorta for which women tended to have greater values. Multivariable regression analysis using age and body size (weight, height, and body surface area) predicted all aortic diameters, whereas blood pressure indexes predicted only the distal part of the aorta. Aortic stiffness increased with age in men and women with no differences between genders; only age predicted aortic stiffness. The increment in aortic diameter with age was lesser when adjusted for aortic stiffness. In conclusion, we define the physiologic range of aortic root diameters and related stiffness in healthy subjects stratified by age and gender. Moreover, aortic stiffness should also be taken into account when the increase of aortic diameter is considered.
The measurement of aortic diameters plays a key role in the clinical evaluation and management of diseases of the aorta. Among cardiovascular imaging techniques, bidimensional Doppler transthoracic echocardiography (TTE), being widely available, cost effective, and safe, represents an excellent screening tool to evaluate aortic root morphology and dimensions. However, data on the normal range of proximal thoracic aortic diameters in subjects free from cardiovascular disease are limited with respect to sample size, measurement sites, and composition of analyzed cohorts. In contrast, aortic diameters alone are not always sufficient to manage aortic disease pathology. Therefore, we might need to take into consideration other parameters to have a better understanding of the prognosis and the treatment of aortic disease.
The aim of the present study was to evaluate by TTE the full spectrum of aortic root diameters and related stiffness in a wide age range of healthy subjects and to investigate clinical and echocardiographic correlates.
Methods
From June 2008 to January 2013, 422 Caucasian volunteers (from 16 to 90 years) without known cardiovascular risk factors and/or overt cardiovascular disease were examined in the echocardiographic laboratory of the department of cardiology, San Antonio Hospital, San Daniele del Friuli (Udine, Italy) for the purpose of the present study. The subjects underwent voluntary full screening for cardiovascular disease, including a questionnaire about medical history, the use of medications, cardiovascular risk factors, and lifestyle habits (alcohol intake, smoking, and physical activity). Physical examination (height, weight, heart rate, and blood pressure [BP]) and clinical assessment were conducted according to standardized protocols by trained and certified staff members. Body surface area (BSA) was calculated with the DuBois formula [0.20247 × height (m) 0.725 × weight (kg) 0.425 ]. Three BP measurements were obtained from the right arm with a mercury manometer. Pulse pressure (PP) was calculated as systolic BP (SBP) − diastolic BP (DBP) and mean BP as DBP − 1/3 PP. The present study was approved by the Institution’s Ethics Board, and informed consent was obtained from the participants.
Standardized TTE examinations were performed using commercially available equipment for all subjects (Aloka α10; Aloka, Tokyo, Japan). All studies were reviewed and analyzed off-line by 2 independent observers (OV and CD). Specific measurements were made using the average of 3 to 5 cardiac cycles. Two-dimensional measurements were performed in the parasternal long-axis view with the patient in the left lateral position. Measurements included left ventricular (LV) internal diameters in diastole and systole and the interventricular septum and the posterior wall in diastole. LV mass was calculated by the Penn convention and indexed by BSA. LV ejection fraction was calculated by Simpson’s rule from the apical 4-chamber view. LV stroke volume (LVSV) was calculated by measuring the LV outflow tract area and the amount of blood going through this area (tracing the signal envelope of the velocity time integral of the pulse Doppler sample in the LV outflow tract). Doppler-derived LV diastolic inflow was recorded in the apical 4-chamber view by placing the sample volume at the tip level of the mitral valve leaflets. LV diastolic measurements included E and A peak velocities (meter/second) and their ratio as well as E-wave deceleration time (milliseconds).
Two-dimensional measurements of the aortic root were made at end-diastole in the parasternal long-axis views at 4 levels (1) the annulus (defined echocardiographically as the hinge points of the aortic cusps), (2) the sinuses of Valsalva, (3) the sinotubular junction, and (4) the proximal ascending aorta. The measurements were made perpendicular to the long axis of the aorta using the leading edge technique in the views showing the largest aortic diameters.
Aortic elasticity was assessed using a 2-dimensional guided M-mode evaluation of the systolic aortic diameter (AoS) and the diastolic aortic diameter (AoD), 3 cm above the aortic valve. AoD was obtained at the peak of the R wave on the simultaneously recorded electrocardiogram, whereas AoS was measured at the maximal anterior motion of the aortic wall. The following indexes of aortic stiffness were calculated: pressure strain elasticity modulus [Ep = (SBP − DBP)/(AoS − AoD)/AoD] and aortic stiffness (β) = ln(SBP/DBP)/[(AoS − AoD)/AoD], where SBP and DBP refer to brachial BP and ln(SBP/DBP) refers to the natural logarithm of the relative pressure. The β stiffness index may be considered relatively unrelated to pressure changes, whereas the Ep is pressure dependent. The study was approved by the Institution’s Ethics Board, and informed consent was obtained from the participants.
Data analysis was performed using Stata, version 10.0 (StataCorp LP, College Station, Texas). Demographics, LV measurements, aortic diameters (absolute and relative to BSA), and stiffness are presented as mean ± SD. The subjects were divided into 5 age groups (ages: 16 to 29, n = 92, 74% men; 30 to 39, n = 76, 64% men; 40 to 49, n = 90, 72% men; 50 to 59, n = 85, 66% men; ≥60, n = 79, 58% men) and then stratified by gender. Difference in gender was tested by the unpaired t test; analysis of variance was performed to evaluate differences within the age groups. The increment in aortic diameter among age groups was calculated and then adjusted by wall thickness. Partial correlation test by Pearson’s method was done to assess clinically relevant variables with p <0.05 and then incorporated into the multivariate model. Five models were used in the multiple regression analysis to explain aortic dimensions. Model A included age and gender; model B included age, gender, and BSA; model C included age, gender, weight, and height; model D included age, gender, BSA, SBP, and mean BP; and model E included age, gender, weight, height, SBP, and mean BP. Also the independent association between aortic stiffness and demographic, anthropometric, clinical, and echocardiographic data was studied. The variables considered were age, gender, body mass index, SBP, mean BP, PP, and LVSV in model A and age, gender, weight, height, SBP, mean BP, PP, and LVSV in model B. Two-tailed p value <0.05 was considered statistically significant. Reproducibility of aortic measurements was determined in 41 subjects. The inter- and intraobserver variability was examined using both Pearson’s bivariate 2-tailed correlations.
Results
The baseline clinical and echocardiographic characteristics of the overall study population are listed in Table 1 . Men were heavier (weight: p <0.001, BSA: p <0.001), taller (p <0.001), and had higher SBP, DBP, and heart rate (p <0.05) than women. They also had greater LV mass index and stroke volume (p <0.001).
| Variable | All Group (n = 422) | Men (n = 284, 67%) | Women (n = 138, 33%) | p |
|---|---|---|---|---|
| Age (yrs) | 44.35 ± 16.91 | 43.08 ± 16.72 | 46.94 ± 17.06 | 0.029 |
| Weight (kg) | 73.0 ± 13.53 | 78.41 ± 11.98 | 61.87 ± 8.98 | <0.001 |
| Height (cm) | 172.64 ± 9.4 | 177.17 ± 6.81 | 163.33 ± 7.0 | <0.001 |
| BSA (m 2 ) | 1.86 ± 0.2 | 1.95 ± 0.17 | 1.67 ± 0.13 | <0.001 |
| BMI (kg/m 2 ) | 24.4 ± 3.5 | 24.9 ± 3.4 | 23.2 ± 3.4 | <0.001 |
| SBP (mm Hg) | 128.04 ± 16.15 | 129.27 ± 15.49 | 125.52 ± 17.21 | 0.031 |
| DBP (mm Hg) | 76.23 ± 10.35 | 77.0 ± 10.41 | 74.67 ± 10.09 | 0.028 |
| Heart rate (beats/min) | 68.15 ± 13.01 | 66.79 ± 13.65 | 70.94 ± 11.13 | 0.001 |
| Mean BP (mm Hg) | 93.50 ± 11.15 | 94.42 ± 10.88 | 91.62 ± 11.49 | 0.018 |
| PP (mm Hg) | 51.78 ± 12.36 | 52.23 ± 12.3 | 50.83 ± 12.49 | NS |
| LV mass index (BSA) (g/m 2 ) | 94.31 ± 25.49 | 99.95 ± 25.30 | 82.75 ± 21.79 | <0.001 |
| SV (ml) | 71.22 ± 18.24 | 76.22 ± 17.85 | 61 ± 14.38 | <0.001 |
| E/A | 1.68 ± 0.81 | 1.68 ± 0.75 | 1.67 ± 0.93 | NS |
The absolute aortic diameter values are reported in Figure 1 . Aortic measurements were significantly greater in men than in women for all age groups. The indexed aortic diameters were similar in both genders except for the annulus and ascending aorta for which women tended to have greater values and also in this case the indexed diameters increased across age groups ( Table 2 ).
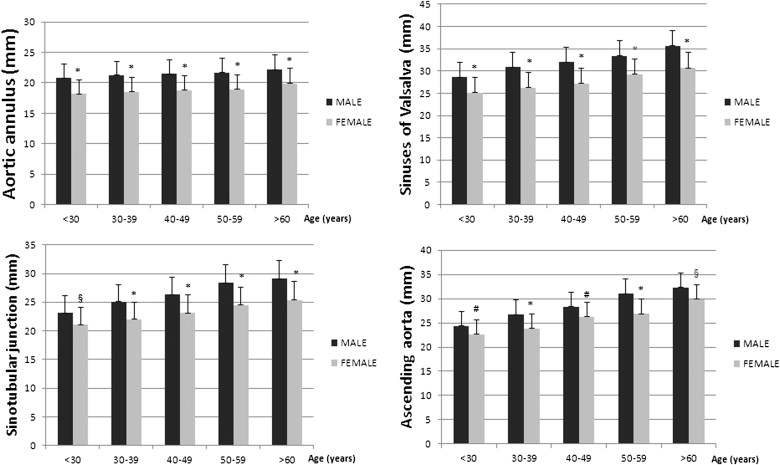
| Age (yrs) | Annulus | Sinuses of Valsalva | Sinotubular Junction | Proximal Ascending Aorta | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| M | W | p | M | W | p | M | W | p | M | W | p | |
| 16–29 (M 68/W 24) | 10.91 ± 1.32 | 10.75 ± 1.1 | NS | 14.98 ± 1.62 | 14.87 ± 1.49 | NS | 12.11 ± 1.55 | 12.5 ± 1.87 | NS | 12.73 ± 2.99 | 13.36 ± 1.71 | NS |
| 30–39 (M 49/W 27) | 10.76 ± 1.312 | 11.35 ± 1.12 | 0.03 | 15.61 ± 1.7 | 16.49 ± 1.88 | NS | 12.69 ± 1.48 | 13.45 ± 1.71 | NS | 13.47 ± 1.23 | 14.71 ± 1.7 | 0.004 |
| 40–49 (M 65/W 25) | 11.08 ± 1.4 | 11.08 ± 1.05 | NS | 16.49 ± 1.9 | 16.06 ± 1.9 | NS | 13.56 ± 1.64 | 13.75 ± 1.7 | NS | 14.56 ± 1.6 | 15.6 ± 2.1 | 0.045 |
| 50–59 (M 56/W 29) | 10.93 ± 1.2 | 11.45 ± 1.1 | 0.05 | 16.86 ± 1.86 | 17.76 ± 2.32 | NS | 14.31 ± 1.76 | 14.84 ± 2.0 | NS | 15.65 ± 1.9 | 16.26 ± 2 | NS |
| ≥60 (M 46/W 33) | 11.65 ± 1.22 | 12.05 ± 1.0 | NS | 18.76 ± 2.5 | 18.55 ± 2.35 | NS | 15.28 ± 2.24 | 15.37 ± 1.86 | NS | 16.92 ± 2.24 | 18.11 ± 2.1 | 0.022 |
| ANOVA among age groups, p | <0.0001 | <0.001 | — | <0.001 | <0.001 | — | <0.001 | <0.001 | — | <0.001 | <0.001 | — |
Aortic diameters were significantly associated with age and gender (model A) and BSA (model B). BP variables were independently associated with the aortic diameters only at the distal part of the thoracic aorta (model C). Weight and height did not have any additional significant impact on aortic dimension (models D and E) ( Table 3 ).
| Variable | Model A R 2 = 0.27 | Model B R 2 = 0.34 | Model C R 2 = 0.34 | Model D R 2 = 0.34 | Model E R 2 = 0.34 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| β | p | β | p | β | p | β | p | β | p | |
| Aortic annulus | ||||||||||
| Age | 0.2 | 0.0001 | 0.2 | 0.0001 | 0.19 | 0.0001 | 0.2 | 0.0001 | 0.10 | 0.0001 |
| Gender | −0.51 | 0.0001 | −0.32 | 0.0001 | −0.32 | 0.0001 | −0.29 | 0.0001 | −0.29 | 0.0001 |
| BSA | 0.299 | 0.0001 | 0.29 | 0.0001 | ||||||
| Weight | 0.21 | 0.0001 | 0.2 | 0.0001 | ||||||
| Height | 0.14 | 0.03 | 0.14 | 0.029 | ||||||
| SBP | 0.049 | NS | 0.05 | NS | ||||||
| MBP | 0.02 | NS | −0.007 | NS | ||||||
| R 2 = 0.46 | R 2 = 0.495 | R 2 = 0.498 | R 2 = 0.50 | R 2 = 0.50 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| β | p | β | p | β | p | β | p | β | p | |
| Sinuses of Valsalva | ||||||||||
| Age | 0.53 | 0.0001 | 0.53 | 0.0001 | 0.5 | 0.0001 | 0.53 | 0.0001 | 0.5 | 0.0001 |
| Gender | −0.48 | 0.0001 | −0.32 | 0.0001 | −0.32 | 0.0001 | −0.29 | 0.0001 | −0.29 | 0.0001 |
| BSA | 0.24 | 0.0001 | 0.23 | 0.0001 | ||||||
| Weight | 0.18 | 0.0001 | 0.17 | 0.001 | ||||||
| Height | 0.12 | 0.027 | 0.13 | 0.024 | ||||||
| SBP | −0.017 | NS | 0.07 | NS | ||||||
| MBP | 0.180 | NS | −0.024 | NS | ||||||
| R 2 = 0.38 | R 2 = 0.425 | R 2 = 0.438 | R 2 = 0.43 | R 2 = 0.44 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| β | p | β | p | β | p | β | p | β | p | |
| Sinotubular junction | ||||||||||
| Age | 0.519 | 0.0001 | 0.52 | 0.0001 | 0.498 | 0.0001 | 0.53 | 0.0001 | 0.51 | 0.0001 |
| Gender | −0.39 | 0.0001 | −0.22 | 0.0001 | −0.21 | 0.0001 | −0.18 | 0.001 | −0.18 | 0.001 |
| BSA | 0.27 | 0.0001 | 0.26 | 0.0001 | ||||||
| Weight | 0.16 | 0.002 | 0.15 | 0.005 | ||||||
| Height | 0.178 | 0.004 | 0.19 | 0.003 | ||||||
| SBP | −0.19 | 0.014 | −0.19 | 0.017 | ||||||
| MBP | 0.24 | 0.002 | 0.24 | 0.002 | ||||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


