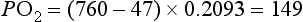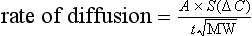Chapter 1
Anatomy and physiology of the respiratory system
This chapter is not intended to provide a comprehensive description of the anatomy and physiology of the respiratory system; rather it is intended to provide a level of understanding that is required to underpin the remainder of this book. Readers wishing to gain a deeper insight into this interesting area are referred to the work of Davies & Moores (2010) and Lumb (2010).
THORACIC STRUCTURE AND FUNCTION
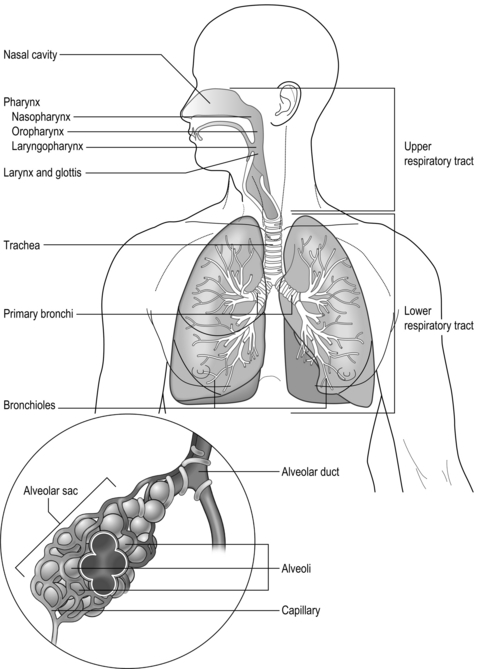
The respiratory system is illustrated in Figure 1.1 and is made up of all the structures that guide air into the lungs (nose, mouth, and airways), plus the lungs themselves and the structures that surround the lungs (thoracic cavity, including the rib cage). The right lung comprises three lobes, whilst the left has two, which allows space for the heart to lie between the left lobes, sloping toward the left. The weight of both adult lungs is between 0.7 and 1.0 kg (1.5 and 2.2 pounds) when weighed at autopsy; however, in life they probably weigh twice this amount because the blood vessels within the lungs (pulmonary circulation) will be filled with about 0.9 litres of blood (weighing about 0.95 kg [2.1 pounds]). In other words, the adult human has about 2 kg (4.4 pounds) of lung tissue hanging inside the rib cage.
The airways
The airways branch a total of 23 times, creating a tree-like structure that ends in the alveoli, where the exchange of oxygen (O2) and carbon dioxide (CO2) takes place (Fig. 1.2). The branches follow an irregular, dichotomous pattern in which each airway gives rise to two ‘daughter’ airways. The structure is irregular because the daughter branches may not be of equal size. The number of airways (N) in each generation (Z) is given by the equation N = Z2.
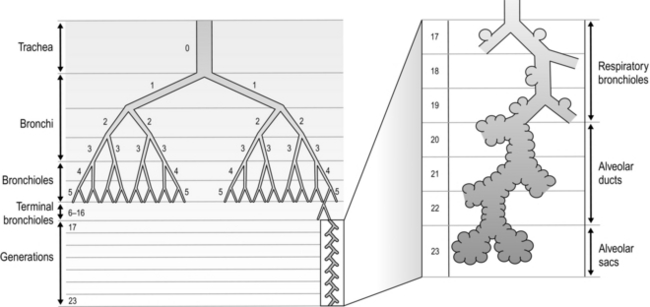
Figure 1.2 Diagram illustrating the branching structure of the lung airways. (With permission from Davies A, Moores C, 2010. The respiratory system. Churchill Livingstone, London, p. 15, Fig. 2.4.)
Air enters via the nose and mouth; then it travels into the pharynx, through the glottis and down the trachea. Next, the air travels into the right and left bronchi, and then through the branching structure of the remaining airways to the alveoli. The alveoli are collections of air sacs, similar to a bunch of grapes, which are surrounded by a dense network of capillaries (think of a bunch of grapes inside a net shopping bag; Fig. 1.1). The regions of the lung without alveoli (including the airways) are known as the conducting zone (branches 1–16), whilst the regions with alveoli are known as the respiratory zone (17–23), i.e., the zone where oxygen and carbon dioxide are exchanged (Fig. 1.2). From branch 17 onwards (respiratory bronchioles), the airways begin to display alveolar buds in their walls, and by branch 20 onwards virtually the entire airway is made up of alveoli (alveolar ducts). An important feature of the conducting airways is that the larger airways, such as the trachea, are reinforced with cartilage rings that help prevent collapse, whereas the walls of smaller airways contain no supporting skeleton. The small airways possess rings of smooth muscle that, when contracted, narrow the airways (bronchoconstriction). From branch 3 onwards, the airways are surrounded by lung parenchyma, and the elastic forces that operate to recoil the lung parenchyma help to tug the airways open during exhalation (airway tethering), with their radial traction (see section ‘Mechanics of breathing: Airway resistance’).
The alveoli
The branching structure of the lungs is an impressive work of evolution that has resulted in adult human lungs having a combined surface area of about 60 m2 (646 square feet), which is about the same as a singles badminton court and about 40 times the area of the skin (see Fig. 1.23). Why the need for such a huge area? Like so much of evolution, the respiratory system is a slave to the laws of physics. As will be described in detail in the next section, the exchange of oxygen (O2) and carbon dioxide (CO2) between the 300 million alveoli and the capillaries surrounding them occurs via passive diffusion. For this process to keep pace with the metabolic needs of the average person, especially during exercise, a vast surface area (number of alveoli and capillaries) is required for diffusion.
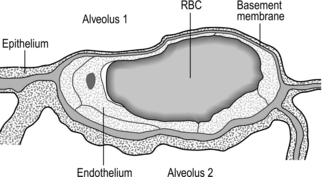
Figure 1.3 Drawing taken from an electron micrograph section of an alveolus showing the alveolar–capillary membrane. RBC = red blood cell. (With permission from Davies A, Moores C, 2010. The respiratory system. Churchill Livingstone, London, p.18, Fig. 2.9.)
However, this vast lung surface area would be of little use without the unique and intimate interrelationship that exists between the alveoli and their capillary network. Figure 1.3 illustrates the key feature of this structure, i.e., the minimal distance separating the alveolar air from the capillary blood. Note that on one side of the capillary the alveolar and capillary cells fuse to form a very thin septum, whilst on the opposite side of the capillary the septum is thicker, providing stability and resisting collapse.
The blood vessels
The second circulation in the lungs arises from the aorta as the bronchial arteries, which meet the metabolic requirements of the conducting airways by perfusing the walls of the airways as far as the respiratory bronchioles (after which O2 requirements are met by alveolar gas exchange). Around one-quarter to one-third of the venous effluent from the bronchial circulation drains into the bronchial veins and thence to the right atrium. The remainder drains directly into the pulmonary veins via bronchopulmonary–arterial anastomoses, contributing to shunting of deoxygenated blood into the pulmonary veins. This shunt is the reason that the alveolar to arterial pressure difference for oxygen exists (see section ‘Gas exchange, Diffusion’ below).
The nerves
The respiratory system is under both somatic and autonomic nervous control. The somatic system provides motor control of respiratory pump muscles, whereas the autonomic system provides both motor (efferent) and sensory (afferent) nerves to the lungs. For information on somatic nerve innervation of respiratory muscles see ‘The muscles’ (below).
The muscles
Respiratory pump muscles
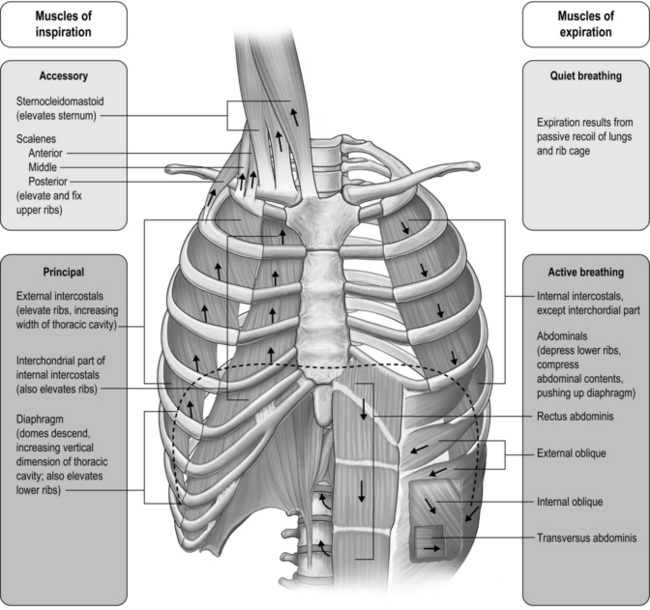
The respiratory pump muscles are a complex arrangement that form a semi-rigid bellows around the lungs. Essentially, all muscles that attach to the rib cage have the potential to generate a breathing action, but the principal muscles are shown in Figure 1.4. Muscles that expand the thoracic cavity are inspiratory muscles and induce inhalation, whereas those that compress the thoracic cavity are expiratory and induce exhalation. These muscles possess exactly the same basic structure as all other skeletal muscles, and they work in concert to expand or compress the thoracic cavity. The structure of the rib cage is described in the section ‘Gross structure of the respiratory system’ (below).
Functional properties of respiratory pump muscles
• Type I: Slow contracting and relatively weak, but very resistant to fatigue
• Type IIA: Moderately fast and strongly contracting, with high resistance to fatigue
• Type IIX (also known as type IIB): Fast and very strong, but with only moderate resistance to fatigue.
It comes as no surprise to learn that the proportion of oxidative fibres (type I and type IIA) within the diaphragm and inspiratory intercostals is approximately 80% (Gollnick et al, 1972), whilst that of the expiratory intercostals is almost 100%. This compares with around 35–45% for limb muscles (Gollnick et al, 1972). The fibres of the abdominal muscles tend to be much more variable in their composition, reflecting the multiplicity of their roles. Another important factor determining muscle fatigue is its blood supply. Inadequate blood flow (ischaemia) not only limits oxygen delivery, it also limits the delivery of substrates and the removal of metabolic by-products, all of which can hasten fatigue. The diaphragm and rib cage muscles are supplied by numerous arteries. The diaphragm, for example, is perfused by three arteries, as well as benefiting from anastamoses that provide collateral sources of blood flow between arteries. The diaphragm also has an extremely dense capillary network and a capacity to increase its blood flow that exceeds that of limb muscles (Polla et al, 2004). The diaphragm maintains perfusion at contraction forces that occlude blood flow in limb muscles. This advantage derives from the fact that it is a thin sheet of muscle that produces a negative intrathoracic pressure during contraction; this pressure gradient maintains blood flow (Buchler et al, 1985). It has been suggested that this abundant and persistent arterial supply protects the diaphragm fibres from ischaemia (Hussain, 1996), providing resistance to fatigue.
In the past, the highly fatigue-resistant characteristics of the respiratory pump muscles contributed to a key assumption regarding the likelihood that the respiratory muscles contributed to exercise limitation. Physiologists assumed that the respiratory pump muscles, especially the diaphragm, were so well evolved from their continuous work that they were immune to fatigue. It wasn’t until the 1990s that this myth was finally shattered, when exercise-induced diaphragm fatigue was measured in healthy young athletes (see Ch. 3, section ‘Respiratory muscle involvement in exercise limitation, Healthy people’).
Upper airway muscles
The first question to address is why upper airway muscles are relevant to breathing. The simple answer is that, without them, upper airway resistance would be intolerable. During normal resting breathing, the vocal folds abduct during inhalation in order to widen the laryngeal glottic opening, permitting unobstructed air flow through the larynx (Brancatisano et al, 1984). This occurs via reflex activation of the posterior cricoarytenoid (PCA) muscle. Without this activity, the vocal folds would collapse across that laryngeal opening, causing an increase in resistance to upper airway flow and leading to increased breathing effort and dyspnoea. The strength of contraction of the PCA muscles has been shown to be proportional to factors that are associated with increased levels of respiratory drive, as well as the negativity of intrathoracic pressure (Suzuki & Kirchner, 1969). During vigorous breathing the action of the PCA is supplemented by contraction of the cricothyroid (CT), which acts to tension the vocal folds, increasing the anteroposterior dimension of the larynx (Hoh, 2005). Active closure of the vocal folds (adduction) is performed by the lateral cricoarytenoid muscle (LCA), thyroarytenoid (TA) and interarytenoid (IA), but only the PCA is involved in resting breathing. However, during tidal breathing most of the closure is brought about by relaxation of the PCA rather than by activation of the adductor muscles (Murakami & Kirchner, 1972). Transient, reflex modulation of the area of the laryngeal portion of the airway plays an important role in controlling breathing frequency, duty cycle and end-expiratory lung volume, as narrowing of the airway provides an important braking effect during expiration. Active adduction is associated with activities such as vocalization, coughing and straining.
The fibre type of human laryngeal muscles has not been studied nearly so extensively as limb muscles, but a number of key observations have been made with respect to the vocal fold abductors (PCA and CT). The PCA and CT contain around 66% and 45% type I fibres, respectively. The type II fibres of these muscles are limited to IIA and IIX, but the latter are very few in number. In common with limb muscles, PCA and CT appear to contain no IIB fibres (Hoh, 2005). The proportion of fast IIX fibres appears to be larger in the adductor muscles (TA and LCA), which probably imparts a higher velocity of shortening. This may be functionally important in their role as protectors of the airway (Li et al, 2004).
Because of its role in vocalization, the LCA has been studied in relation to its fatigability. Intramuscular EMG (electromyography) suggests that prolonged, loud vocalization exercises result in changes within the EMG that are consistent with the development of muscle fatigue (Boucher et al, 2006). Thus, it is reasonable to suggest that under similarly challenging conditions for abduction (e.g., vigorous breathing) PCA and CT might be similarly susceptible to fatigue. This is discussed in relation to exercise intolerance in Chapter 3 (section ‘Respiratory muscle involvement in exercise limitation’).
Gross structure of the respiratory system
The respiratory system is housed within the thoracic cavity, which is formed by the rib cage, vertebrae, sternum and diaphragm. Within the thorax there are three further cavities: the left and right pleural cavities and the mediastinum (Fig. 1.5).
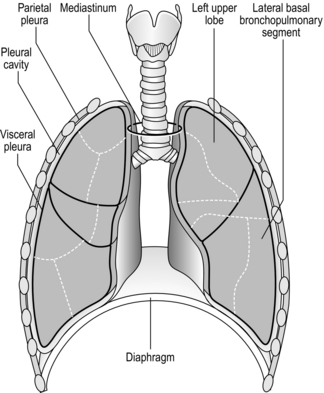
Figure 1.5 Gross anatomy of the lungs. (With permission from Davies A, Moores C, 2010. The respiratory system. Churchill Livingstone, London, p. 21, Fig. 2.12.)
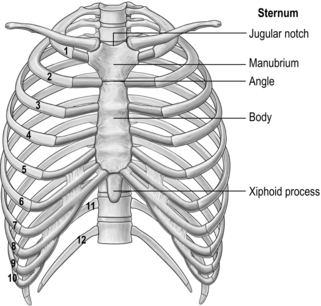
The arrangement and movements generated by the respiratory muscles are described above (in ‘The muscles’). The skeletal structures that translate the muscle actions into movements are complex. The rib cage structure is such that the vertebral articulation of the ribs is higher than the sternal attachments; consequently, the ribs slope downwards anteriorly (Fig. 1.6).
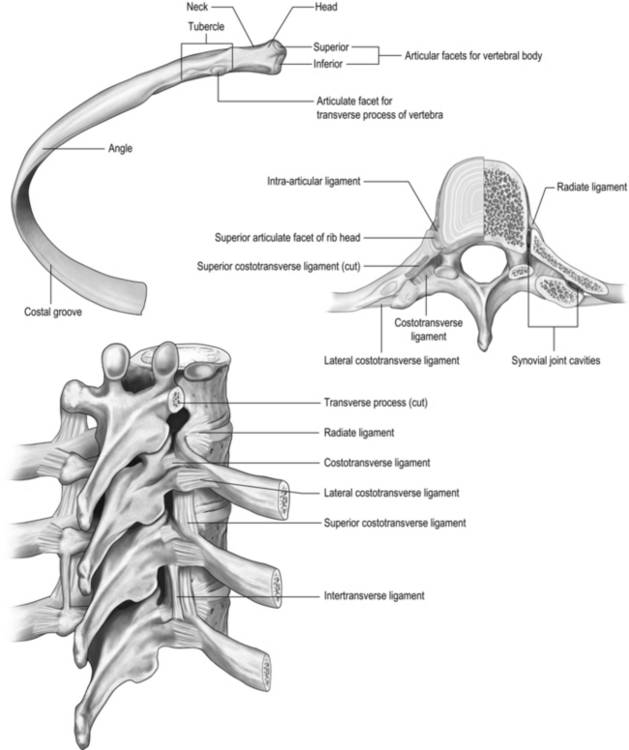
All 12 ribs articulate with the vertebrae, but only the first six connect individually to the sternum. Ribs 7–10 articulate with the lower sternum via a common cartilage, whilst ribs 11 and 12 have no anterior connections (‘floating ribs’). The head of most ribs articulates with the bodies of two adjacent vertebrae, whilst their tubercles articulate with the transverse process of one of these vertebrae. Nerves and blood vessels run in a channel on the underside of the ribs called the costal groove (Fig. 1.7).
GAS EXCHANGE
However, the partial pressure of oxygen within the alveoli (PAO2) is not 149 mmHg because the inhaled breath is diluted by the air that remained in the lungs at the end of the previous breath (end-expiratory lung volume, EELV). The extent of the dilution depends upon a number of factors, including the EELV and the tidal volume (VT); typically PAO2 will be around 104 mmHg, giving a diffusion gradient of around 65 mmHg. Thus, VT affects both the flow of gas delivered to the alveoli (via the dead space to VT ratio; see ‘Lung volumes and capacities’, below), and the extent to which inspired PO2 (PIO2) is diluted by the EELV (inspired to alveolar gradient). This mixing of old and new gas also affects the diffusion gradient and exchange of CO2 because it has the effect of raising the partial pressure of CO2 (PCO2) in the alveoli (PACO2). Thus, it is easy to see how changes in breathing pattern can influence the driving pressure for gas exchange, leading to hypoxaemia and hypercapnia.
where A is the diffusion area, S is the solubility coefficient of the gas, ΔC is concentration gradient for the gas (alveolar to pulmonary arterial), t is the membrane thickness (alveolar plus capillary), and MW is the molecular weight of the gas molecule (its physical size). Although CO2 is 23 times more soluble than O2, it has a larger MW making the actual diffusion rate 20 times greater. However, equilibrium between the alveolus and venous blood occurs at around the same time for both CO2 and O2 because of the lower ΔC and slower release of CO2 from the blood compared with that of O2. Importantly, the A in the Fick equation is not the lung surface area, but rather the surface area where there is both ventilation and perfusion, i.e., where air and blood meet. As will be explained in later sections, many factors can influence the distribution of both ventilation and perfusion, leading to ventilation / perfusion (![]() /
/ ![]() ) inequality, a reduction in A and an impairment of diffusion.
) inequality, a reduction in A and an impairment of diffusion.
There is one final factor that influences the efficiency of gas exchange between alveolus and blood, viz., the characteristics of the blood supply to the alveolus, and in particular the time available for the diffusion and binding / release of O2 and CO2. Figure 1.8 is a diagrammatic illustration of the change in pulmonary blood gases as the blood traverses the lungs. Under resting conditions, the blood spends around 0.75 seconds in contact with the alveolar regions of the lung, during which time it will traverse a number of alveoli.
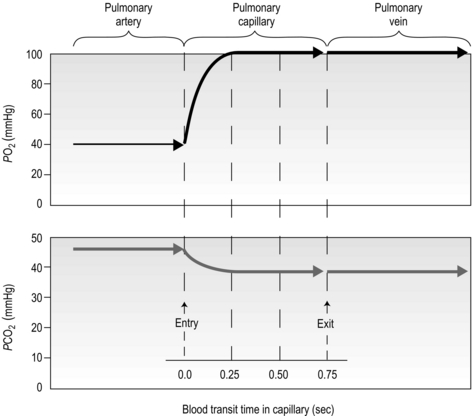
Figure 1.8 Equilibrium time for O2 and CO2 as blood traverses the alveolar capillaries under resting conditions. (With permission from Hicks GH, 2000. Cardiopulmonary anatomy and physiology. WB Saunders, Philadelphia, p. 365, Fig. 12-17.)
It is clear from Figure 1.8 that equilibration of both gases occurs early in the journey through the capillary, and that within 0.25 seconds diffusion ceases because there is no longer a gradient and exchange is complete. The only way to increase the exchange of gas is to increase the throughput of blood. In this scenario, gas exchange is perfusion limited. Many people find it hard to understand that, in healthy people at sea level, breathing more during exercise does not improve the amount of oxygen leaving the lungs; this is because it’s already ‘as good as it gets’. However, diffusion limitation can arise in healthy people under very specific conditions including: (1) exposure to high altitude (reducing the diffusion gradient), (2) during maximal exercise, when (a) cardiac output can be so high that blood traverses the lung capillaries before full equilibration can take place, (b) cardiac output can be so high that it outstrips the ability of the respiratory pump to deliver adequate alveolar ventilation (VA), (c) ![]() /
/ ![]() inequality develops due to the mechanical effects of acute pulmonary oedema compressing alveoli and capillaries, and (d) the diffusion distance is increased by acute pulmonary oedema. Exercise-induced arterial hypoxaemia in healthy well-trained individuals at sea level is relatively rare and the specific contributions of the putative contributor remains incompletely understood (Guenette & Sheel, 2007).
inequality develops due to the mechanical effects of acute pulmonary oedema compressing alveoli and capillaries, and (d) the diffusion distance is increased by acute pulmonary oedema. Exercise-induced arterial hypoxaemia in healthy well-trained individuals at sea level is relatively rare and the specific contributions of the putative contributor remains incompletely understood (Guenette & Sheel, 2007).
Perfusion
The pulmonary vasculature is supplied via a separate, low-pressure branch of the cardiovascular system. Deoxygenated blood is returned to the lungs via the right side of the heart and the pulmonary artery. The latter is the only artery in the body to carry deoxygenated blood, which is distributed to a huge capillary network within the lungs (Fig. 1.9).
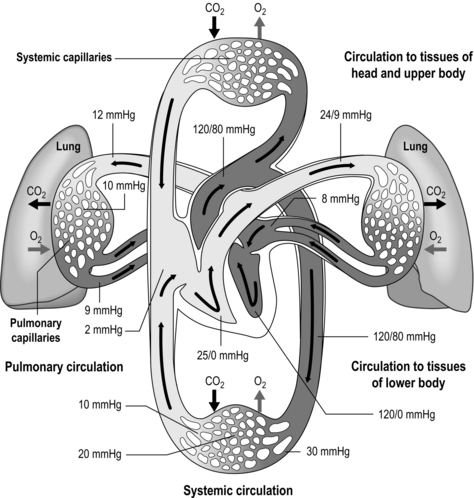
Figure 1.9 Schematic depicting pulmonary and systemic circulations and pressures. (With permission from Thibodeau GA, Patton KT, 1995. Anatomy and physiology, 3rd edn, Mosby, St Louis.)
As well as illustrating the flow of blood through both branches of the circulation, the diagram in Figure 1.9 also illustrates the pressures present within each part. Systolic / diastolic pressure in the pulmonary artery is around only 24 / 9 mmHg, compared with 120 / 80 mmHg in the aorta. The pulmonary circuit is at low pressure because of the thin, delicate walls of the capillary network, which minimizes the diffusion distance. Even small increases in pulmonary arterial pressure lead to fluid leakage and pulmonary oedema. Since the pulmonary circulation receives virtually the entire cardiac output, the fact that the pressures within it are low indicates that it has a low resistance. The reason for this low resistance is the extensiveness of its capillary network; as was described above, the capillary network can be thought of as a sheet of blood enveloping the alveoli.
One factor that has a strong influence upon the patency of pulmonary capillaries is alveolar pressure, which can compress the vessels. This increases their resistance, leading to redistribution of blood and heterogeneity of blood flow within the lungs. In addition, gravity, which also influences alveolar pressure (see ‘Pressures within the thorax’, below), has a potent influence upon blood flow distribution within the lungs. The effect is due to the hydrostatic pressure gradient that exists from the apex to the base of the lungs. It is very potent because pulmonary arterial pressure is so low that it is only just sufficient to pump blood to the lung apices. Perfusion pressure at the base of the lungs is equivalent to pulmonary arterial pressure, plus the hydrostatic pressure difference between the heart and base of the lungs. At the apex, perfusion pressure is equivalent to pulmonary arterial pressure, minus the hydrostatic pressure difference between the heart and apex of the lungs. This means that blood flow is distributed preferentially to the base of the lungs. As will be described in a later section (‘Pressures within the thorax’), ventilation is also distributed preferentially to the base of the lungs. However, the relative distribution of ventilation and perfusion is imperfect; there is around a three- to four-fold difference in ventilation from apex to base, compared with only a 16-fold difference in perfusion. Accordingly, there is a gradient of ventilation perfusion ratios (![]() /
/ ![]() ), and gas exchange, throughout the lungs. These variations in gas exchange also lead to local variations in the alveolar gas partial pressures. These phenomena are summarized in Figure 1.10.
), and gas exchange, throughout the lungs. These variations in gas exchange also lead to local variations in the alveolar gas partial pressures. These phenomena are summarized in Figure 1.10.
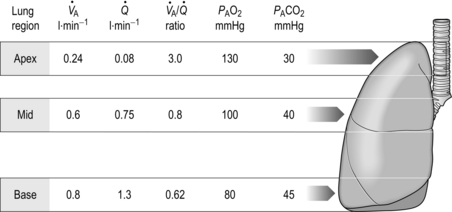
Figure 1.10 Variations in alveolar ventilation (![]() A) and perfusion (
A) and perfusion (![]() ). (With permission from Hicks GH, 2000. Cardiopulmonary anatomy and physiology. WB Saunders, Philadelphia, p. 352, Fig. 12-9.)
). (With permission from Hicks GH, 2000. Cardiopulmonary anatomy and physiology. WB Saunders, Philadelphia, p. 352, Fig. 12-9.)
In summary, the factors influencing blood-flow distribution in the lungs include:
• Gravity (via alveolar pressure and hydrostatic pressure)
• Pulmonary arterial resistance
• Lung volume (via alveolar pressure)
• Alveolar gas pressure (influenced by lung volume and gravity).
• Hypoxic pulmonary vasoconstriction (HPV) is a unique response to hypoxia; in systemic blood vessels, hypoxia elicits vasodilatation. The logic of the differing responses is obvious since there is no point in perfusing areas of the lung that are poorly ventilated. In contrast, in the periphery, hypoxic tissue requires an increase in oxygen delivery and dilatation is the way to achieve this. The effect of HPV is to direct blood to ventilated regions, thereby improving ![]() /
/ ![]() for the lung as a whole. In patients with conditions such as COPD (chronic obstructive pulmonary disease), in whom alveolar hypoxia is a chronic phenomenon, HPV can result in extensive pulmonary vasoconstriction. This increases PVR and the load placed upon the right side of the heart. As a result, both pulmonary oedema and right heart failure (cor pulmonale) can ensue.
for the lung as a whole. In patients with conditions such as COPD (chronic obstructive pulmonary disease), in whom alveolar hypoxia is a chronic phenomenon, HPV can result in extensive pulmonary vasoconstriction. This increases PVR and the load placed upon the right side of the heart. As a result, both pulmonary oedema and right heart failure (cor pulmonale) can ensue.
• The pulmonary circulation is also influenced by a number of endogenous substances that induce vasodilatation via nitric oxide (NO) release by pulmonary vessel endothelial cells. These substances include acetylcholine, bradykinin, thrombin, serotonin, adenosine diphosphate and histamine. In addition, mechanical factors such as stretching and vessel wall shear stress may also induce release of NO and vasodilatation.
• The pulmonary vasculature falls under the influence of the autonomic nervous system. Sympathetic stimulation releases norepinephrine (noradrenaline), which stimulates α1-receptors in the smooth muscle of pulmonary arteries and arterioles, inducing vasoconstriction. The parasympathetic nervous system releases acetylcholine, which induces vasodilatation via the NO system described above. A recent study suggests that sympathoexcitation due to muscle metaboreflex activation induces pulmonary vasoconstriction (Lykidis et al, 2008), which has implications for gas exchange and exercise tolerance.