Fig. 11.1
The influence of the neuroendocrine system on the immune system. The symbols + and − indicate activation and inhibition, respectively. MT melatonin, PRL prolactin, E estrogen, T testosterone, OT oxytocin, MSH melanocyte stimulating hormone, GH/iGF-1 growth hormone/insulin-like growth factor-1, MIF macrophage inhibitory factor
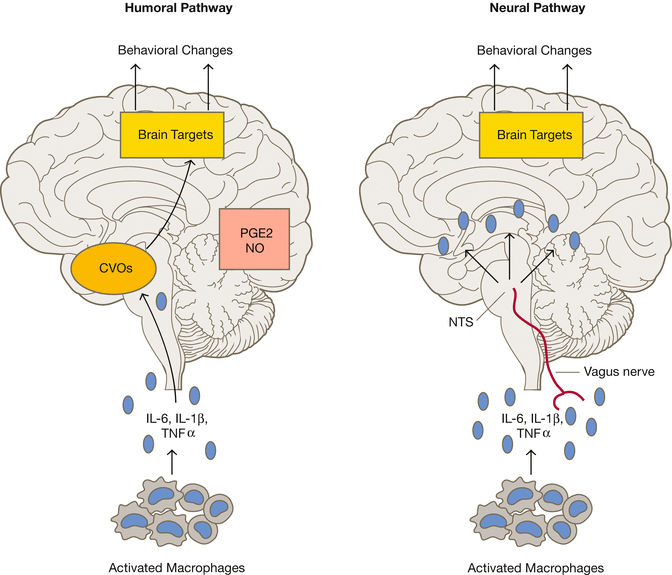
Fig. 11.2
Cytokines from immune cells activated (i.e., macrophage) reach the brain via the blood and the vagus nerve. Via the blood they reach the blood–brain barrier-free circumventricular organs (CVO) and elicit a second messenger (prostaglandins, nitric oxide) in blood–brain barrier too; via the vagus nerve they reach the nucleus tractus solitarius, the brain stem integration centre
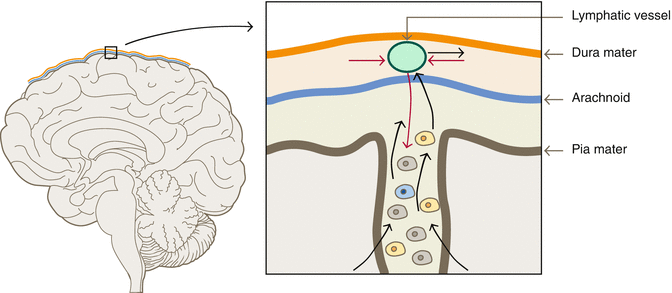
Fig. 11.3
The discovery of the lymphatic vessel [16] shows a new bidirectional pathway from the brain to the immune system
At the same time, it has by now become clear how cytokines, which are released by the immune cells, pass through the blood or the large cranial nerves (such as the vagus nerve) and carry signals to the brain, thus influencing both biological (e.g., fever, hunger, satiety) and psychological activities (anxiety and depression) [11, 12].
During the 1990s, there was a significant boom in studies of the neurobiology of emotions. Emotions, traumas, and stressful events in general cause a dysregulation in the stress system that strongly alters the disposition and the functioning of the immune system. In the short-term, cortisol, epinephrine, and norepinephrine (NE) (catecholamines) also have a tonifying effect on the immune system. In the mid- to long-term, these substances place the immune response in an unsuitable position to fight viruses and tumors. Similarly, the dysregulation of the stress axis can favor the onset of different types of autoimmune diseases [13].
By the end of the twentieth century, the works of the American neuroscientist Robert Sapolsky and those of other scholars have proven that the alteration of the stress system and the overproduction of cortisol can result in atrophy of the hippocampus, the cerebral area responsible for long-term memory formation.
Studies of the first decade of the twenty-first century show that even diseases such as atherosclerosis and heart disease are usually strongly affected by mood. Depression, with the overproduction of cortisol and catecholamines, contributes to alterations in the inner vessel walls, favoring the formation of atherosclerotic lesions and worsening the prognosis of people who have suffered a heart attack [14]. Thus, the occurrence of some heart attacks or other acute cardiac failures tied to mood disorders, can be correlated with the presence of vascular alterations caused by an imbalance in serotonin levels, as suggested from animal research [15]. Serotonin has multiple physiological effects: in the brain the humor is regulated; in the gut, it is peristaltic activity; and in the blood it is coagulation that is regulated. Its decrement in the brain triggers depressive effects, whereas increment in the blood produces increased coagulation (prothrombotic effects).
To conclude, in the first decade of this century, research conducted mainly by the Belgian psychiatrist Michael Maes and the French neurobiologist Robert Dantzer proved that immune inflammatory dysregulation may be responsible for symptoms that are normally referred to as “somatization disorders” and “psychosomatic symptoms.” All of these symptoms are tied to disorders that fall either within the purview of psychology and psychiatry (anxiety, depression, chronic fatigue syndrome) or to disorders belonging more specifically to the medical field (autoimmune diseases, cancer). For a recent general review, it is possible to consult the article by Irwin and Rothermund [17].
With PNEI, a model for research and for interpreting both health and disease has developed, one that considers the human body to be a structured and interconnected entity where the psychological and biological systems have a reciprocal influence. This provides the basis for new integrated approaches to the prevention and therapy of most common diseases, especially chronic ones. At the same time, it affords us the prospect of going beyond the old philosophical dichotomy between mind and body and the scientific dichotomy between medicine and psychology, by overcoming the respective reductionism that assigns the body (without the psyche) to the care of medicine, and the psyche (without the body) to that of psychology.
11.2 The Science of Stress
Research into PNEI has evolved out of the experimental pathological studies by Hungarian scientist Hans Selye, rightly considered the father of stress science.
From the 1930s until his death in 1982, Hans Selye focused his research on the adaptation of human and animal organisms in response to different kinds of stressors (physical, psychological, and toxic agents). Hence, the subject of Selye’s investigations was the living organism as a whole. This approach to the unity of the organism was based on the experimental observation that the axis of stress in animals was activated independently of the nature of the stressor. The adrenal axis of an experimental mouse could be activated by a virus, a bath in icy water, or the animal’s catching sight of a predator. This is now well documented. When bacteria or a virus get into the body, our immune system releases cytokines that reach the brain and activate the stress system. This same stress system can also be activated by an emotion. Today we know that this “nonspecificity” of stimuli is also present at the cellular level. Indeed, an immune cell can be activated by viral and bacterial products, inflammatory cytokines, oxidative derivatives in food, or by emotional stress: the cell has receptors for epinephrine and NE, which are the neurotransmitters of the stress response. Therefore, even if it seems inappropriate to presuppose a basic difference between psychological and biological stressors, there is undoubtedly a distinctiveness intrinsic to the psychological stressors.
Here is the fundamental question: how does the psyche work? What are the mechanisms by which an internal or external event activates the stress system?
11.3 The Psyche as a Product and Condition of the Life of the Organism
The psyche is an integral part of the meta-system. It is both the product of the biological organization and a fundamental source of its conditioning. It is necessary to fully understand that the biological and psychological levels are inextricably entwined. If we miss this point, we will be faced with two diverging philosophical trajectories: biologism and subjective idealism. The former considers the psyche nothing more than a synonym for the brain and the latter simply recycles the old metaphysical concept of Soul or Spirit. As a matter of fact, the psyche is an integral and vital part of the body.
A recent review lists the identified correlations between behaviors and brain plasticity (e.g., from navigation and hippocampus, from music and motor and auditory areas) and subsequently recalls humorously the famous conundrum: “what came first, the chicken or the egg?” [18].
Hand in hand with these correlations between brain structure and behaviors, a series of experiments has also hypothesized about the molecular paths that convert behaviors into a new cerebral asset, showing the increase in signals that are essential to the activation and growth of nerves such as brain derived nervous factor (BDNF), nervous growth factor (NGF), n-methyl- d -aspartate (NMDA).
Perspective-based parallel studies have been carried out. In these studies, researchers monitored people in the process of acquiring new knowledge and skills over a period of time.
In an experiment, they involved German medical students. At the end of their third academic year, students are required to pass an extremely demanding state medical examination, the so-called Physicum, a summary of all the main subjects studied in the previous years. This challenging examination requires on average a 2-month period of intense study. In this case, as in the other, the participants’ brains were imaged three times, first before the study period, and then after, at the 2-month and 3-month marks. Neuroimages showed significant growth and an increase in grey matter in two crucial learning areas: the posterior parietal cortex and the hippocampus. Of particular relevance is the fact that after 3 months the hippocampus, especially the right side, continued to grow as if it were further processing the acquired information [19]. One may conclude from this that mental activity modifies brain morphology, regardless of whether it involves acquisition of abstract notions or acquisition of motor skills.
We can therefore make the following analogy: the software running on the brain machine modifies the machine itself.
For this reason, the psyche–brain system cannot be compared with the program computer system. In this latter case, if one changes the software, the hardware does not follow suit, whereas in the first case (the psyche–brain system), the software modifies the hardware.
11.4 Psycho-neuro-endocrino-immunology, the Science of Stress, and the Biopsychosocial Model
From the arguments and scientific data reported thus far, it can be deduced that it is now possible to unify the two great traditions (the biological and the psychological) of stress research, with the aim of reconstructing the health/disease balance and the mechanisms that concern the individual seen in his/her entirety. What is needed is a new unified science of stress.
Scholars of psychological sciences respond to this assertion by arguing that for some time there has been a systemic approach, called the “biopsychosocial model.”
This proposal was published nearly 40 years ago in Science [20], where the author of the article, George Engel, did indeed present a very effective criticism of the dominant biomedical model by identifying its key elements: the reduction of complex phenomena to simple determinants (reductionism), the separation of the biological phenomena from the psychosocial ones (mind–body dichotomy), and the interpretation of vital phenomena in physical–chemical terms (physicalism).
Unfortunately, the course of history has not quite matched the vision set forth by Engel. Admittedly, the biopsychosocial model has trumpeted the idea, over the last few decades, that another vision of medicine and psychology is possible, but in all honesty, it has not advanced any research that explores the mechanisms contributing to the interdependence of the biological, the psychological, and the social. Thanks to PNEI we now have all the tools, as well as a rich and growing body of documentation, necessary for explaining and defining, in strictly scientific terms, the complex interaction between the different levels that determine human health and disease.
It is therefore no coincidence that distinguished researchers in psychological sciences, such as Shelley Taylor and Janice Kiecolt-Glaser, see PNEI as the forward path of choice for psychology. Kiecolt-Glaser writes that “Psychoneuroimmunology is psychology’s gateway to the biomedical future.” Indeed, it represents the principal contribution, she writes, that psychology can make in the effort to change the biomedical model and practice. It is her belief that to achieve this goal, an interdisciplinary framework for the training of health operators and researchers is necessary [21].
11.5 PNEI and Cardiovascular Diseases
Cardiovascular mortality and morbidity, and the problem of gender differences in cardiovascular diseases (CVDs) are relevant problems in world-wide disease epidemiology.
In the USA, the 2010 overall rate of death attributable to CVDs was 235.5 per 100,000. The rates were 278.4 per 100,000 for white males, 369.2 per 100,000 for black males, 192.2 per 100,000 for white females, and 260.5 per 100,000 for black females. From 2007 to 2010, 40.0% of men and 34.4% of women aged 40–59 suffered from CVD in the USA, and these percentages rise to 70.2 and 70.9% for ages 60–79 and 83.0% and 87.1% for ages 80 and above [22].
Working from a life-course perspective, Liu and Waite [23] have formulated hypotheses about age and gender differences in the link between marital quality and cardiovascular risk. They have tested these hypotheses using data from the first two waves of the National Social Life, Health, and Aging Project. The analysis sample includes 459 married women and 739 married men (aged 57–85 in the first series) who were interviewed in both periods. Results suggest that changes in marital quality and cardiovascular risk are more closely related for older married people than for their younger counterparts; and that the link between marital quality and cardiovascular risk is more pronounced among women than among men of older age. These findings fit with the gendered life-course perspective and the cumulative disadvantage framework.
In this disadvantage framework, depression and stress play an important role.
Recently, in the second wave of the Nord-Trøndelag Health Study in Norway (HUNT 2, 1995–1997), baseline data on symptoms of anxiety and depression, along with health status including cardiovascular risk factors, were registered for 62,567 adults, men and women, who were free for known indicators for heart failure. This group was monitored for incident heart failure (HF) from baseline throughout 2008. Symptoms of depression were associated with an increased risk for heart failure (HF) in a dose–response manner [24].
On 24 February 2014, the American Heart Association, with a scientific statement, determined that depression is a risk factor with a poor prognosis among patients with acute coronary syndrome [25]. With this report, we have for the first time unequivocal testimony of the negative influence that depression has on the progression of CVD. What are the possible biological mechanisms at work here?
11.6 Biological Mechanisms 1: Autonomic Cardiac System Dysregulation
Several forebrain areas form an interconnected network that initiates integrated autonomic, endocrine, and behavioral changes in response to emotionally relevant or homeostatically stressful stimuli [26]. This central stress network, including the insular cortex (IC), anterior cingulate cortex (ACC), amygdala (A), and several hypothalamic nuclei, projects to medullary and spinal nuclei controlling cardiac function by the vagus and sympathetic nerves.
This circuit appears essential to the regulation of autonomic responses to stressful stimuli. An imbalance in autonomic regulation may be linked to mental stress, resulting in a net shift toward increased sympathetic tone and/or withdrawal of parasympathetic tone. If chronic, this imbalance can increase the risk for cardiovascular disease by promoting arrhythmias, platelet aggregation, and vascular inflammation.
The sympathetic innervations of the heart originate in a group of neurons of the intermediolateral column (IML) of the spinal cord, which receives tonic excitatory glutamatergic inputs from neurons in rostral ventrolateral medulla (RVLM). The parasympathetic innervations of the heart are provided by the vagus nerve, by cardiovagal neurons primarily located in the nucleus ambiguus (NAmb) and in the minor extension in the dorsal motor nucleus of the vagus (DMN).
The autonomic cardiac system influences the intrinsic cardiac nervous system, a complex neural network composed of ganglia embedded in epicardial fat pads and the heart wall.
The sinoatrial (SA) node and atrioventricular (AV) node are two internal pacemakers that are responsible for initiating the heartbeat.
Sympathetic activation, via noradrenergic neurons, elicits an increase in the automatism of the SA node, with an increase in the heart rate (HR), and in AV conduction; in contrast, the main effects of the vagus, via cholinergic neurons, are the inhibition of the pacemaker activity of the SA node (decrease HR), and reduced AV conduction.
11.6.1 Arrhythmias and Autonomic Nervous System
It is well recognized that sympathetic inputs represent major neural triggers for both atrial and ventricular arrhythmias [27].
Atrial arrhythmia (atrial fibrillation and atrial flutter the most observed) is the most common rhythm disorder: the prevalence is increasing and accounts for nearly one quarter of ischemic strokes in the elderly population.
Ventricular arrhythmia is the leading cause of sudden death, a serious public health problem with 200,000–450,000 events in the USA annually [28]. About 50% of deaths in patients with heart failure are due to fatal arrhythmias.
Experimentally, sympathetic stimulation induces a change in ECG repolarization and reduces the fibrillation threshold, facilitating the initiation of ventricular fibrillation [29]. These effects are magnified in the presence of ischemic damage to the myocardial and conducting tissue.
Zhou et al. [30] demonstrated that ventricular fibrillation and sudden cardiac death, in ambulatory dogs, are immediately preceded by spontaneous sympathetic nerve discharge, indicating that sympathetic nerve discharge may serve as a trigger for arrhythmia.
A non-invasive marker of vulnerability to life-threatening arrhythmias in cardiovascular disease patients is heart rate variability (HRV); in general, reduced HRV indicates excessive cardiac sympathetic activity, reduced vagal modulation, and an increased risk of arrhythmic events.
Stress, through its impact on the autonomic cardiac system, can trigger both ventricular and atrial arrhythmias, as demonstrated by epidemiological, clinical, and laboratory studies.
Kop and collaborators [31] reported that mental stress can induce changes in T wave alternans (a sign of electrical instability) occurring at lower heart rates than with exercise in patients with implantable cardioverter defibrillators.
Other studies suggest that patients with heart failure experience alterations in autonomic nervous system function through increased sympathetic outflow to the heart and the peripheral vasculature and decreased parasympathetic activity, as documented by higher plasma NE concentrations and decreased clearance of NE from the circulation due to low cardiac output. Furthermore, increased plasma NE concentrations appear to predict negative outcomes in patients with congestive heart failure (CHF) [32].
In recent years, myocardial infarction with normal coronary arteries (MINCA) has been increasingly recognized and diagnosed. Recently, it was shown that MINCA occurs more frequently than previously thought, and data indicate that the prevalence ranges between 5 and 25% of all myocardial infarctions and approximately one-third of MINCA consists of Takotsubo stress cardiomyopathy.
Takotsubo cardiomyopathy (TTC) is characterized by transient systolic and diastolic left ventricular dysfunction with a variety of wall-motion abnormalities classified as apical, midventricular, basal or focal, in patients without coronary artery disease. It predominantly affects elderly women (except for Japan, where TTC is more prevalent among men) and is often preceded by an emotional trigger, although recent studies indicate that the disease may also occur with physical triggers or even without any evident preceding trigger.
Notably, 15.3% of patients with TTC have evidence of coexisting coronary artery disease on angiography.
The etiology of TTC is unknown, although indications point toward a strong relationship between TTC and acute physiological or mental stress with catecholamine excess and vasospasm.
Takotsubo cardiomyopathy is not an entirely benign and reversible condition. Data from the International Takotsubo Registry, a consortium of 26 centers in Europe and the USA, indicate that rates of severe in-hospital complications including shock and death are similar to acute coronary syndrome and younger patients with physical triggers and acute neurological or psychiatric diseases have a higher incidence of acute complications compared with elderly patients with emotional triggers. Ten-year follow-up combined end points of death and major adverse cardiac and cerebrovascular events reveal a rate of death from any cause of 5.6% per patient-year and a rate of major adverse cardiac and cerebrovascular events of 9.9% per patient-year among TTC patients. In both acute phase and long-term follow-up, men were at a higher risk for major adverse cardiac and cerebrovascular events than women [33].
11.6.2 The Role of the Vagus Nerve
In all the disorders mentioned above, the dysregulation of the autonomic cardiac system is characterized by sympathetic–vagal imbalance with parasympathetic withdrawal.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


